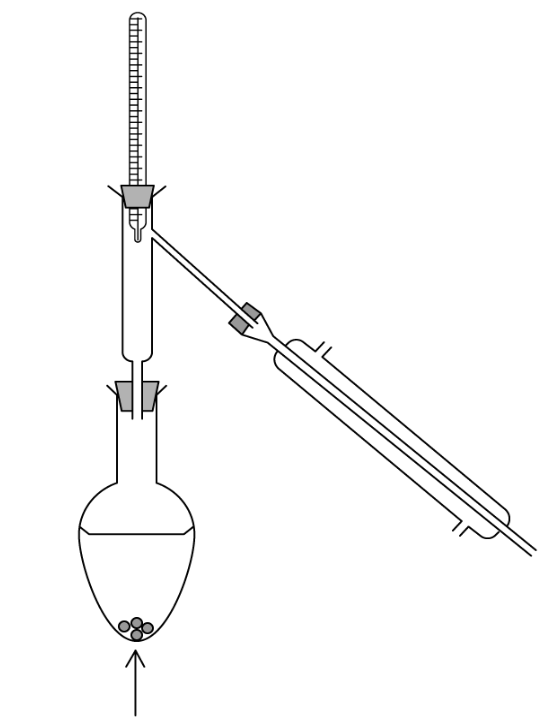
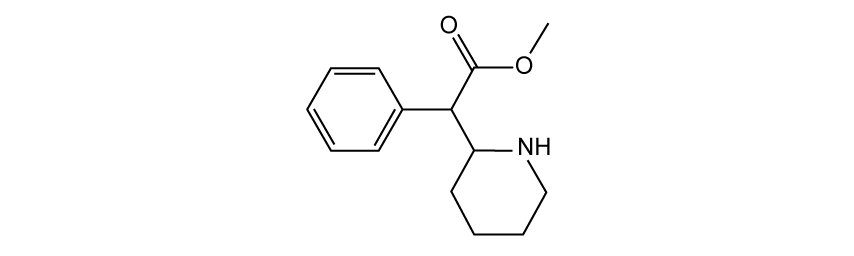
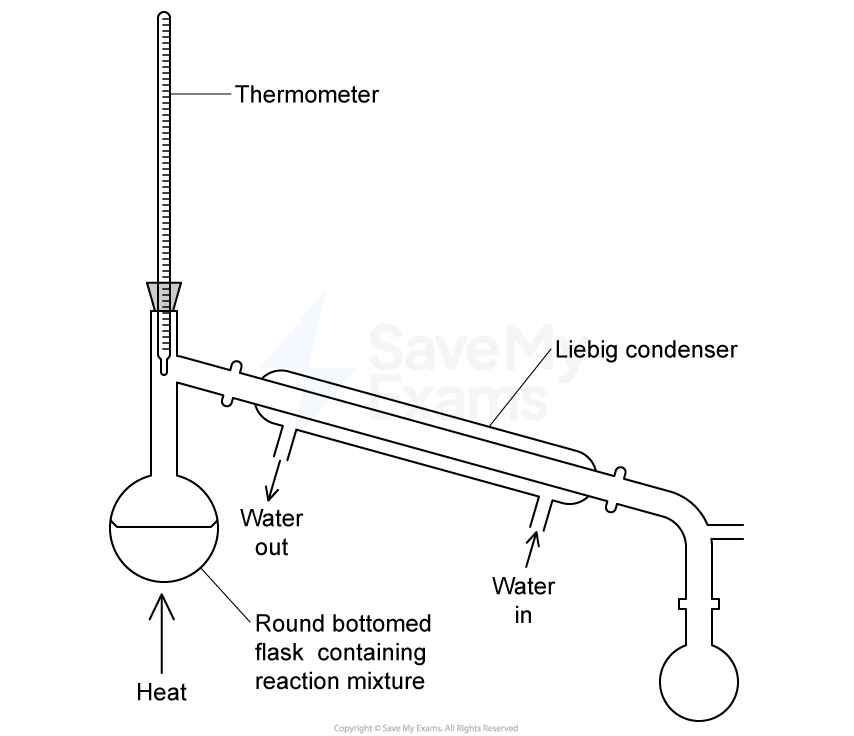
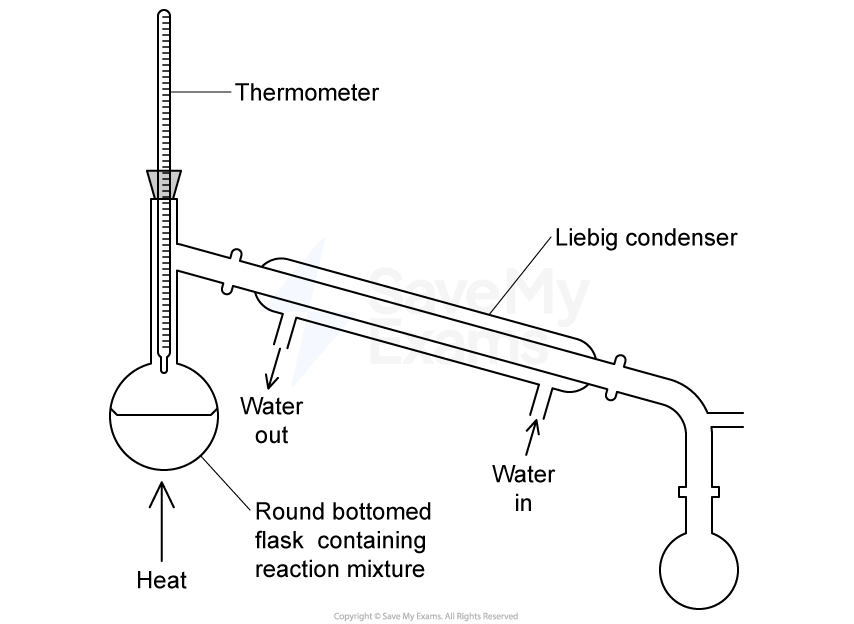
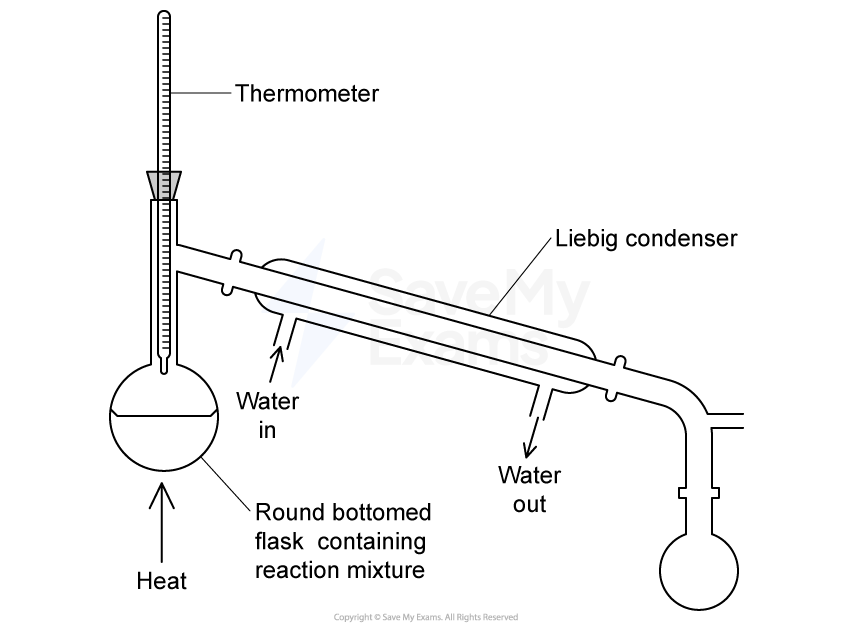
Which statement about heating under reflux is correct?
Organic liquids can be heated without evaporation taking place.
Organic liquids can be maintained at their boiling point for extended periods.
Heating is carried out using a water bath.
The apparatus is closed to prevent the loss of any materials.
Was this exam question helpful?