Which organic compound has the lowest boiling point?
2,3,4-trimethylpentane
2,3-dimethylhexane
2-methylheptane
Octane
Was this exam question helpful?
Exam code: H432
Select a download format for Alkanes
Select an answer set to view for
Alkanes
Which organic compound has the lowest boiling point?
2,3,4-trimethylpentane
2,3-dimethylhexane
2-methylheptane
Octane
Choose your answer
Was this exam question helpful?
In the presence of ultraviolet light, ethane and chlorine react to give a mixture of products.
Which compound could be present in the mixture of products?
CH3CH2CH2CH2CH3
CH3CH2CH2CH3
CH3CH2CH2Cl
CH3Cl
Choose your answer
Was this exam question helpful?
Ultraviolet light initiates the following reaction.
alkane + chlorine → chloroalkane + hydrogen chloride
What happens to chlorine in this photochemical reaction?
Heterolytic fission to give an electrophile
Homolytic fission to give an electrophile
Heterolytic fission to give a positive and negative ion
Homolytic fission to give free radicals
Choose your answer
Was this exam question helpful?
Which equation is correct for the complete combustion of heptane?
2C7H16 + 22O2 → 14CO2 + 16H2O
2C7H16 + 15O2 → 14CO + 16H2O
2C7H14 + 21O2 → 14CO2 + 14H2O
2C6H14 + 19O2 → 12CO2 + 14H2O
Choose your answer
Was this exam question helpful?
Ethane reacts with bromine in the presence of ultraviolet radiation via a radical substitution mechanism.
C2H6 + Br2 → C2H5Br + HBr
Which equation shows a termination step for this reaction?
C2H5● + Br2 → C2H5Br + Br●
C2H5● + Br• → C2H5Br
Br2 → 2Br●
C2H6 + Br• → C2H5• + HBr
Choose your answer
Was this exam question helpful?
Which statement about methane is not correct?
It is a saturated hydrocarbon
It follows the general formula CnH2n
It has a tetrahedral shape
It can undergo free radical substitution
Choose your answer
Was this exam question helpful?
Give the bond angle present in methane.
90°
107°
109.5°
120°
Choose your answer
Was this exam question helpful?
Which row shows possible products from the incomplete combustion of an alkane?
Carbon | Carbon monoxide | Hydrogen | |
|---|---|---|---|
A | ✓ | ✓ | × |
B | ✓ | × | ✓ |
C | × | ✓ | ✓ |
D | ✓ | ✓ | ✓ |
Choose your answer
Was this exam question helpful?
Which statement explains the trend in boiling points of alkanes as carbon-chain length increases?
Covalent bonds become stronger
Hydrogen bonds become stronger
Induced dipole–dipole interactions (London forces) become stronger
Permanent dipole–dipole interactions become stronger
Choose your answer
Was this exam question helpful?
Which statement(s) about propane is/are correct?
It is non-polar.
It cannot undergo incomplete combustion.
It decolourises bromine.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Ethane reacts with bromine in the presence of ultraviolet radiation to form a mixture of organic products.
Which equation shows a propagation step in the mechanism for this reaction?
C2H6 + Br• → C2H5Br + H•
C2H5• + Br• → C2H5Br
C2H5• + Br2 → C2H5Br + Br•
C2H5Br + H• → C2H5• + HBr
Choose your answer
Was this exam question helpful?
Isooctane (2,2,4-trimethylpentane) can be added to fuels for improved performance.
Which is the correct equation for the complete combustion of isooctane?
C8H18 + 8½O2 → 8CO + 9H2O
C8H18 + 12½O2 → 8CO2 + 9H2O
C7H16 + 11O2 → 7CO2 + 8H2O
C8H18 + 13O2 → 8CO2 + 9H2O
Choose your answer
Was this exam question helpful?
1 mol of a compound reacts with 9.5 mol O2 for complete combustion.
What is the formula of the compound?
C6H14
C6H13OH
C7H16
C7H15OH
Choose your answer
Was this exam question helpful?
Which property explains the low reactivity of alkanes?
High C–C bond enthalpy
High polarity of the C-H bond
Electron pair repulsion between σ-bonds
Free rotation about σ-bonds
Choose your answer
Was this exam question helpful?
Butane reacts with bromine in the presence of ultraviolet radiation to form 1-bromobutane.
Which radical(s) is/are present in the mechanism for this reaction?
CH3CH2CH2CH2•
Br•
H•
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Propane reacts with chlorine in the presence of ultraviolet radiation.
What is the overall equation for this reaction?
CH3CH=CH2 + Cl2 → CH3CHClCH2Cl
CH3CH2CH2• + Cl2 → CH3CH2CH2Cl + Cl•
CH3CH2CH2• + CH3CH2CH2• → CH3CH2CH2CH2CH2CH3
CH3CH2CH3 + Cl2 → CH3CH2CH2Cl + HCl
Choose your answer
Was this exam question helpful?
An alkane contains 38 hydrogen atoms per molecule.
What is the empirical formula of the compound?
C18H38
C19H38
C9H19
C9H18
Choose your answer
Was this exam question helpful?
The structure of 2-methylbutane is shown below.
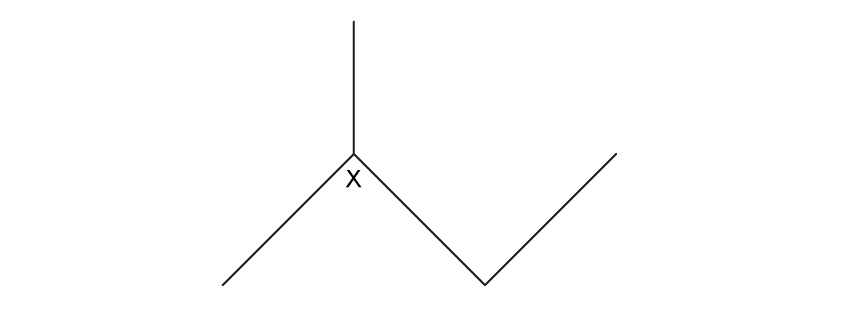
What is the shape around the carbon atom labelled X?
Trigonal planar
V-shaped
Pyramidal
Tetrahedral
Choose your answer
Was this exam question helpful?
Which alkane has the highest boiling point?
CH3CH3
CH3CH2CH2CH3
CH3C(CH3)2C(CH3)2CH3
CH3(CH2 )6CH3
Choose your answer
Was this exam question helpful?
Which substance is not a product of the incomplete combustion of an alkane?
H2O
C
CO
SO2
Choose your answer
Was this exam question helpful?
Chlorine reacts with 1-chloropropane in the presence of ultraviolet radiation via a radical substitution mechanism.
Which equation shows a propagation step in the mechanism for this reaction?
Cl2 → •Cl + •Cl
•Cl + •C3H6Cl → C3H6Cl2
C3H7Cl + •Cl → C3H6Cl2 + •H
•Cl + C3H7Cl → •C3H6Cl + HCl
Choose your answer
Was this exam question helpful?
Which sequence shows these compounds in order of increasing boiling point?
2,4-dimethylheptane
Nonane
Octan-1-ol
Heptanoic acid
2,4-dimethylheptane < nonane < octan-1-ol < heptanoic acid
2,4-dimethylheptane < nonane < heptanoic acid < octan-1-ol
nonane < 2,4-dimethylheptane < octan-1-ol < heptanoic acid
octan-1-ol < heptanoic acid < 2,4-dimethylheptane < nonane
Choose your answer
Was this exam question helpful?
The CFC CCl2F2 can cause the breakdown of ozone in the upper atmosphere.
Which statement(s) about this process is/are correct?
•ClO + O → •Cl + O2 is a propagation step in the breakdown of ozone.
The radicals produced from the initiation of this CFC are •Cl and •F.
An equal concentration of •Cl and •F radicals are produced during initiation.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Chlorine reacts with fluoromethane to form trichlorofluoromethane.
Which statement(s) is/are correct?
The first propagation stage is CH3F + •Cl → •CH2F + HCl, followed by •CH2F + Cl2 → CH2ClF + •Cl.
A termination step in this reaction will produce a product with the empirical formula CHCl2F.
A limitation of this reaction is that further substitution occurs, leading to a mixture of organic products.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
The equation for the incomplete combustion of heptane is shown below.
C7H16 (g) + 8O2 (g) → 6CO (g) + CO2 (g) + 8H2O (g)
Average bond enthalpies are given in the table.
Bond | Average bond enthalpy / kJ mol-1 |
|---|---|
C–C | 348 |
C–H | 414 |
O=O | 498 |
C=O (in CO2) | 804 |
C≡O (in CO) | 1070 |
O–H | 463 |
What is the enthalpy change, in kJ mol-1, for this reaction?
+2740 kJ mol-1
-2740 kJ mol-1
+4474 kJ mol-1
-4474 kJ mol-1
Choose your answer
Was this exam question helpful?