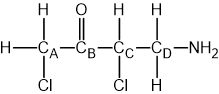
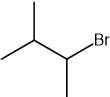
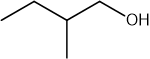
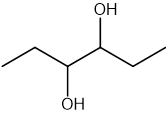
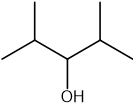
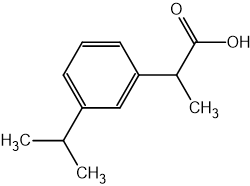
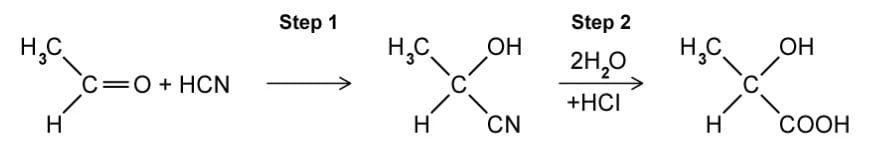
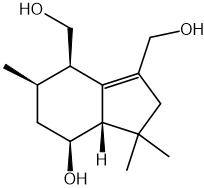
Which statement about optical isomers is correct?
Four different atoms or groups of atoms around a carbon atom cause optical isomerism
Optical isomerism occurs as a result of restricted rotation about a carbon–carbon double bond
Optical isomers are superimposable mirror images of each other called enantiomers
Optical isomers are molecules that have the same structural formula but have the atoms arranged differently in space
Was this exam question helpful?