Which statement about the equation q = mcΔT is correct?
The unit of q is kJ
m represents the amount of substance in moles
c is the specific heat capacity of the substance
The temperature is measured in Centigrade
Was this exam question helpful?
Exam code: H432
Select a download format for Enthalpy Changes
Select an answer set to view for
Enthalpy Changes
Which statement about the equation q = mcΔT is correct?
The unit of q is kJ
m represents the amount of substance in moles
c is the specific heat capacity of the substance
The temperature is measured in Centigrade
Choose your answer
Was this exam question helpful?
Which equation represents both a standard enthalpy change of formation and a standard enthalpy change of combustion?
2CH4 (g) + 3O2 (g) → 2CO (g) + 4H2O (l)
Na (s) + O2 (g) → NaO2 (s)
HCl (aq) + NaOH (aq) → NaCl (aq) + H2O (l)
CO2 (g) + C (s) → 2CO (g)
Choose your answer
Was this exam question helpful?
Which processes have a negative enthalpy change?
The combustion of an alcohol
The reaction between aqueous hydrochloric acid and aqueous sodium hydroxide
The condensation of water vapour
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
The incomplete combustion of methanol can produce carbon monoxide and water.
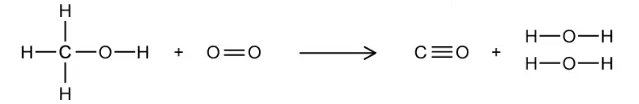
Some average bond enthalpy data is given in the table.
Bond | C–H | C–O | O–H | O=O | C≡O |
|---|---|---|---|---|---|
Average bond enthalpy / kJ mol-1 | 412 | 360 | 463 | 496 | 1077 |
What is the enthalpy change for this incomplete combustion of methanol, in kJ mol-1?
+374
−374
−837
+552
Choose your answer
Was this exam question helpful?
Which is the correct definition for the standard enthalpy of combustion?
The enthalpy change when the reactants in a stoichiometric equation react to give the products, under standard conditions
The enthalpy change when one mole of a substance is burnt in excess oxygen, under standard conditions
The enthalpy change when one mole of water is formed by reacting an acid and an alkali, under standard conditions
The enthalpy change when one mole of a product is formed from its elements, under standard conditions
Choose your answer
Was this exam question helpful?
25.0 cm3 of aqueous nitric acid is reacted with 25.0 cm3 of aqueous potassium hydroxide. The temperature change for the reaction is 7.200 K.
Assume that the density and specific heat capacity, c, of the solutions are the same as for water.
How much energy is released in this experiment?
752 J
752 kJ
1505 J
1505 kJ
Choose your answer
Was this exam question helpful?
The combustion of 1.20 g of ethanol releases 35 500 J of energy.
What is the enthalpy change of combustion of ethanol, in kJ mol-1?
−1 360 000
−29 600
−1360
−29.6
Choose your answer
Was this exam question helpful?
The following Hess cycle can be used to calculate the standard enthalpy change of reaction for the hydrogenation of ethene.

The standard enthalpy changes of combustion, ΔHcθ, are given in the table.
Substance | C~2~H~4~ (g) | H~2~ (g) | C~2~H~6~ (g) |
|---|---|---|---|
ΔHcθ / kJ mol-1 | −1411 | −286 | −1560 |
Which row shows the correct labels for the arrows in the Hess cycle?
Arrow 1 | Arrow 2 | Arrow 3 | Arrow 4 | |
|---|---|---|---|---|
A | ΔHrθ | −1411 | −286 | −1560 |
B | ΔHcθ | −1411 | +286 | −1560 |
C | ΔHcθ | +1411 | −286 | +1560 |
D | ΔHrθ | −1411 | −286 | +1560 |
Choose your answer
Was this exam question helpful?
Which statement explains the term average bond enthalpy?
The energy required to break a specific covalent bond in the gas phase
The energy required to break one mole of a specific covalent bond with all substances in their standard states
The energy required to break a specific covalent bond with all substances in their standard states
The enthalpy change for the breaking of 1 mole of a specified type of bond in gaseous molecules
Choose your answer
Was this exam question helpful?
An enthalpy profile diagram is shown below.

Which row is correct?
Label X | Label Y | Type of reaction | |
|---|---|---|---|
A | Activation energy, Ea | Enthalpy change, ΔH | Exothermic |
B | Enthalpy change, ΔH | Activation energy, Ea | Exothermic |
C | Activation energy, Ea | Enthalpy change, ΔH | Endothermic |
D | Enthalpy change, ΔH | Activation energy, Ea | Endothermic |
Choose your answer
Was this exam question helpful?
Which enthalpy change(s) is / are endothermic?
The bond enthalpy of the C–H bond
The second electron affinity of oxygen
The standard enthalpy change of formation of magnesium
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Enthalpy values are provided below.
H2 (g) + I2 (g) → 2HI (g) ΔrH = −9 kJ mol–1
Bond | Bond enthalpy / kJ mol^-1^ |
|---|---|
H–H | +436 |
I–I | +151 |
What is the bond enthalpy, in kJ mol-1, of the H–I bond?
-596
-298
+298
+596
Choose your answer
Was this exam question helpful?
The table shows standard enthalpy changes of combustion, ΔcH.
Substance | Δ~c~*H* / kJ mol^-1^ |
|---|---|
C (s) | –393.5 |
H2 (g) | –285.8 |
C4H10 (g) | –2876.5 |
What is the enthalpy change for the following reaction?
4C (s) + 5H2 (g) → C4H10 (g)
–2197.2 kJ mol–1
–126.5 kJ mol–1
+126.5 kJ mol–1
+2197.2 kJ mol–1
Choose your answer
Was this exam question helpful?
The table below shows standard enthalpy changes of formation, ΔfH.
Compound | NH4NO3 (s) | H2O (g) | CO2 (g) |
|---|---|---|---|
ΔfH / kJ mol-1 | -366 | -242 | -394 |
What is the enthalpy change for the following reaction?
2NH4NO3 (s) + C (s) → 2N2 (g) + 4H2O (g) + CO2 (g)
–630 kJ mol–1
–96 kJ mol–1
+96 kJ mol–1
+630 kJ mol–1
Choose your answer
Was this exam question helpful?
The table below shows enthalpy changes of formation, ΔfH.
Compound | TiCl4 (l) | H2O (l) | TiO2 (s) | HCl (g) |
|---|---|---|---|---|
ΔfH / kJ mol-1 | -804 | -286 | -945 | -92 |
What is the value of the enthalpy change of reaction, ∆rH, for the reaction in the following equation?
TiCl4 (l) + 2H2O (l) → TiO2 (s) + 4HCl (g)
–63 kJ mol–1
–53 kJ mol–1
+53 kJ mol–1
+63 kJ mol–1
Choose your answer
Was this exam question helpful?
Four possible reactions (A, B, C and D) of the following equation are measured at the same temperature.
X (g) + Y (g) → Z (g)
Which enthalpy profile diagram represents a rapid reaction with a negative enthalpy change?




Choose your answer
Was this exam question helpful?
The table shows the possible signs of three types of standard enthalpy change.
'+' means only positive values are possible.
'–' means only negative values are possible.
'+ or –' means both positive and negative values are possible.
Which row is correct?
| ΔfHθ | ΔcHθ | ΔneutHθ |
|---|---|---|---|
A | + | + | + or – |
B | + or – | + | + or – |
C | + or – | – | – |
D | – | – | + |
Choose your answer
Was this exam question helpful?
A student carried out an experiment to determine the enthalpy change for the combustion of ethanol. In the experiment, ethanol in a spirit burner was ignited and used to heat water in a copper calorimeter.
The specific heat capacity of water is 4.18 J g−1 K−1.
Initial temperature of the water | 21.0 °C |
|---|---|
Final temperature of the water | 54.0 °C |
Mass of water in the calorimeter | 100 g |
Mass of spirit burner before burning | 259.75 g |
Mass of spirit burner after burning | 259.18 g |
What is the energy transferred to the water, in kJ?
8.78
13.8
35.75
13800
Choose your answer
Was this exam question helpful?
The table shows standard enthalpy changes of combustion, ΔcH.
Substance | ΔcH / kJ mol-1 |
|---|---|
H2 (g) | −286 |
C (s) | −394 |
C2H5OH (l) | −1367 |
Calculate the standard enthalpy change of formation, ΔfH, of ethanol.
2C (s) + 3H2 (g) + ½O2 (g) → C2H5OH (l)
687 kJ mol-1
- 279 kJ mol-1
- 2047 kJ mol-1
- 3013 kJ mol-1
Choose your answer
Was this exam question helpful?
The equation for the reaction of nitrogen and fluorine is shown below.
N2 (g) + 3F2 (g) → 2NF3 (g) ΔrH = −246 kJ mol-1
Average bond enthalpies are shown in the table.
Bond | Average bond enthalpy / kJ mol-1 |
|---|---|
N≡N | +945 |
F–F | +159 |
What is the average bond enthalpy, in kJ mol-1, of the N–F bond?
225 kJ mol-1
278 kJ mol-1
1350 kJ mol-1
1668 kJ mol-1
Choose your answer
Was this exam question helpful?
The table shows standard enthalpy changes of formation, ΔfHθ.
Substance | Al2S3 (s) | O2 (g) | Al2O3 (s) | SO2 (g) |
|---|---|---|---|---|
ΔfH / kJ mol-1 | -723.8 | 0 | -1675.7 | -296.8 |
What is the standard enthalpy change of combustion of Al2S3 (s), in kJ mol-1?
-3684.6
-1842.3
+1842.3
+3684.6
Choose your answer
Was this exam question helpful?
The table shows standard enthalpy changes of formation, ΔfHθ.
Substance | SO2 (g) | SO3 (g) |
|---|---|---|
ΔfHθ / kJ mol-1 | −297 | −395 |
What is the enthalpy change, in kJ mol-1, for the following reaction?
2SO2 (g) + O2 (g) → 2SO3 (g)
+196 kJ mol–1
+98 kJ mol–1
-98 kJ mol–1
-196 kJ mol–1
Choose your answer
Was this exam question helpful?
25.0 cm3 of aqueous hydrochloric acid is mixed with 25.0 cm3 of aqueous sodium hydroxide. The initial temperature of both solutions is 18.0 °C. The maximum temperature of the mixture reaches 24.4 °C.
Assume that the density of all solutions and the specific heat capacity, c, of the reaction mixture are the same as for water.
How much energy is released, in J?
669 J
1338 J
2676 J
5100 J
Choose your answer
Was this exam question helpful?
A student carries out an experiment to determine the specific heat capacity of aluminium. The energy absorbed by a 2.5 kg solid aluminium block is 0.1 MJ. The temperature of the block increases by 44.4 °C.
Which of the following gives the specific heat capacity of aluminium to the appropriate number of significant figures and in correct standard form?
9.01 x 102 J K−1 g−1
9.01 x 10-4 J K−1 g−1
9.01 x 10-1 J K−1 g−1
9.01 x 10-7 J K−1 g−1
Choose your answer
Was this exam question helpful?
The table shows the standard enthalpy of formation, ΔHfθ, for some of the substances in the reaction.
4FeS2 (s) + 11O2 (g) → 2Fe2O3 (s) + 8SO2 (g) ΔHrθ = -3312 kJ mol-1
| FeS2 (s) | Fe2O3 (s) |
|---|---|---|
ΔHfθ / kJ mol-1 | -178 | -824 |
What is the standard enthalpy of formation, in kJ mol-1, for SO2 (g)?
-2376 kJ mol-1
-952 kJ mol-1
-297 kJ mol-1
-119 kJ mol-1
Choose your answer
Was this exam question helpful?
A student carries out an experiment to determine the enthalpy change of combustion of a fuel. The student's calculated enthalpy change is less exothermic than the value in data books.
Which of the following is not a reason for this difference?
Heat gained from the surroundings
Heat loss to the surroundings
Evaporation of water
Incomplete combustion
Choose your answer
Was this exam question helpful?
The equation for the complete combustion of heptathiepane, S7, is shown below.
S7 (s) + 7O2 (g) → 7SO2 (g)
Which enthalpy change(s) does this equation represent?
The standard enthalpy change of reaction, ΔHrθ
The standard enthalpy change of combustion of heptathiepane, ΔHcθ
The standard enthalpy change of formation of sulfur dioxide, ΔHfθ
1 | 2 | 3 | |
|---|---|---|---|
A | ✓ | ✓ | ✓ |
B | ✓ | ✓ | ✗ |
C | ✗ | ✓ | ✓ |
D | ✓ | ✗ | ✗ |
Choose your answer
Was this exam question helpful?
The enthalpy changes, ΔHr, for three reactions are shown below.
C2H4 (g) + 2O2 (g) → 2CO (g) + 2H2O (g) ΔHr = −758 kJ mol-1
2C (s) + 2H2 (g) → C2H4 (g) ΔHr = +52 kJ mol-1
2C (s) + O2 (g) → 2CO (g) ΔHr = −222 kJ mol-1
What is the enthalpy change of formation of H2O (g), in kJ mol-1?
-524 kJ mol-1
-484 kJ mol-1
-262 kJ mol−1
-242 kJ mol−1
Choose your answer
Was this exam question helpful?
Butane can be produced by the hydrogenation of buta-1,3-diene.
C4H6 (g) + 2H2 (g) → C4H10 (g) ΔHr = −248 kJ mol-1
Some average bond enthalpy data are given in the table.
Bond | C–C | C–H | H–H |
|---|---|---|---|
Average bond enthalpy / kJ mol-1 | 348 | 412 | 436 |
Use the information in the table and the stated enthalpy change to calculate the average bond enthalpy for the C=C bond in buta-1,3-diene.
786 kJ mol-1
612 kJ mol-1
1224 kJ mol-1
830 kJ mol-1
Choose your answer
Was this exam question helpful?
In the gas phase, phosphorus pentachloride can be thermally decomposed into gaseous phosphorus trichloride and chlorine.
PCl5 (g) → PCl3 (g) + Cl2 (g)
Bond enthalpies are shown in the table.
Bond | P–Cl (in both chlorides) | Cl–Cl |
|---|---|---|
Bond enthalpy / kJ mol-1 | 328 | 241 |
What is the enthalpy change of the reaction, in kJ mol-1?
–415 kJ mol–1
+415 kJ mol–1
95 kJ mol–1
–95 kJ mol–1
Choose your answer
Was this exam question helpful?