Which molecule is not planar?
C2H4
C2H6
H2CO
HCN
Was this exam question helpful?
Exam code: H432
Select a download format for The Shapes of Simple Molecules & Ions
Select an answer set to view for
The Shapes of Simple Molecules & Ions
Which molecule is not planar?
C2H4
C2H6
H2CO
HCN
Choose your answer
Was this exam question helpful?
Which element has induced dipole–dipole interactions (London forces) in its solid lattice?
Boron
Magnesium
Silicon
Sulfur
Choose your answer
Was this exam question helpful?
What is the shape around the carbon atoms in graphene?
Linear
Pyramidal
Tetrahedral
Trigonal planar
Choose your answer
Was this exam question helpful?
A ‘dot-and-cross’ diagram for nitrogen trichloride, NCl3, is shown below.
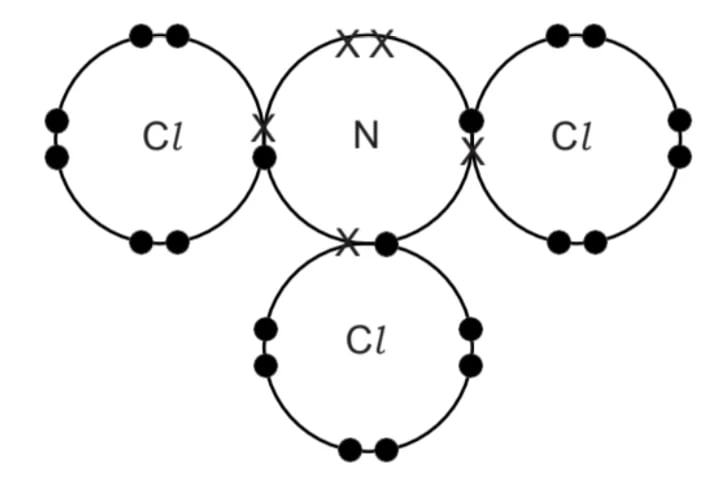
Which row shows the correct shape and bond angle in a molecule of NCl3?
| Name of shape | Bond angle |
|---|---|---|
A | Pyramidal | 104.5 ° |
B | Pyramidal | 107 ° |
C | Tetrahedral | 107 ° |
D | Trigonal planar | 120 ° |
How did you do?
Was this exam question helpful?
Which molecule is non-polar?
SF6
H2O
PF3
NH3
Choose your answer
Was this exam question helpful?
Which substance contains hydrogen bonding in the liquid state?
CH3(CH2)4CH3
CH3(CH2)3CHFCH3
CH3(CH2)3COCH3
CH3(CH2)3CH(OH)CH3
Choose your answer
Was this exam question helpful?
The boiling point of hydrogen bromide is –67 °C.
The boiling point of hydrogen iodide is –34 °C.
The different boiling points can be explained in terms of the strength of bonds or interactions.
Which bonds or interactions are responsible for the higher boiling point of hydrogen iodide?
Covalent bonds
Hydrogen bonds
Induced dipole–dipole interactions
Permanent dipole–dipole interactions
Choose your answer
Was this exam question helpful?
What is the bond angle in a trigonal planar molecule?
107°
120°
180°
109.5°
Choose your answer
Was this exam question helpful?
Which row is correct for an SF6 molecule?
Shape | Bond angle | |
|---|---|---|
A | Octahedral | 90° |
B | Square planar | 90° |
C | Trigonal bipyramidal | 120° |
D | Tetrahedral | 109.5° |
Choose your answer
Was this exam question helpful?
Which row is correct for a molecule of CCl4?
Total number of valence electrons | Number of bonding pairs | Number of lone pairs | |
|---|---|---|---|
A | 30 | 4 | 2 |
B | 32 | 4 | 0 |
C | 34 | 4 | 1 |
D | 16 | 4 | 0 |
Choose your answer
Was this exam question helpful?
What is the shape and bond angle of a molecule of hydrogen sulfide, H2S?
Linear, 180o
Non-linear, 104.5o
Tetrahedral, 109.5o
Trigonal planar, 120o
Choose your answer
Was this exam question helpful?
What is the shape and bond angle of the phosphonium ion, PH4+?
Tetrahedral and 109.5°
Square planar and 90°
V-shaped and 105°
Tetrahedral and 107°
Choose your answer
Was this exam question helpful?
Which molecule is nonpolar?
CH3CHICH3
BF3
BrF5
CH3CHO
Choose your answer
Was this exam question helpful?
Which one of these species has a bond angle of 120°?
H3O+
TlBr32-
BCl3
NH3
Choose your answer
Was this exam question helpful?
What is the strongest type of intermolecular force in the amino acid molecule serine?
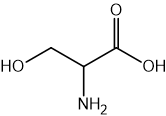
London dispersion forces
Permanent dipole-dipole forces
Hydrogen bonding
Covalent bonding
Choose your answer
Was this exam question helpful?
Which organic compound has the highest boiling point?
Butan-1-ol
Butane
1-chlorobutane
Propane
Choose your answer
Was this exam question helpful?
Which molecule has the lowest boiling point?
CH3CH2CHO
CH3CH2CH2Cl
CH3CH2CH3
CH3CH2COOH
Choose your answer
Was this exam question helpful?
Why is water more dense than ice?
Ice contains hydrogen bonds, but water does not contain hydrogen bonds
Hydrogen bonds are stronger in water than in ice
Hydrogen bonds hold H2O molecules closer together in water
Ice is a solid and water is a liquid
Choose your answer
Was this exam question helpful?
Why does hexane have a higher boiling point than 2,2-dimethylbutane?
Hexane has a higher Mr than 2,2-dimethylbutane
2,2-dimethylbutane has a lower boiling point as it is a branched molecule
2,2-dimethylbutane only contains London dispersion forces
Hexane contains permanent dipole dipole forces
Choose your answer
Was this exam question helpful?
Which element has induced dipole dipole interactions (London dispersion forces) in its solid lattice?
Sodium
Diamond
Phosphorus
Beryllium
Choose your answer
Was this exam question helpful?
Four atoms, A-D, are labelled in the structure of alanine below.
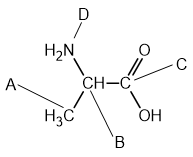
Which atom has a trigonal planar arrangement of bonds around it?
Choose your answer
Was this exam question helpful?
Which compound has polar molecules?
OCl2
BCl3
BCl4
SCl6
Choose your answer
Was this exam question helpful?
The boiling point of butan-1-ol is 118 °C. The boiling point of 2-methylpropan-2-ol is 82 °C.
Why is the boiling point of butan-1-ol higher than that of 2-methylpropan-2-ol?
Butan-1-ol has stronger induced dipole–dipole interactions because it has more electrons
Butan-1-ol has stronger induced dipole–dipole interactions because it has a straight-chain structure
Butan-1-ol can form hydrogen bonds while 2-methylpropan-2-ol cannot
Butan-1-ol is more stable because it is a primary alcohol
Choose your answer
Was this exam question helpful?
Which statement correctly explains the trend in the boiling points of the halogens from chlorine to iodine?
The boiling point increases because the permanent dipole-dipole interactions become stronger as the molecules become more polar.
The boiling point increases because the number of electrons increases, leading to stronger London (induced dipole-dipole) forces.
The boiling point decreases because the covalent bond strength decreases as the atomic radius increases.
The boiling point decreases because the shielding effect increases, weakening the attraction between molecules.
Choose your answer
Was this exam question helpful?
Which compound has non-polar molecules?
E-1,2-dichlorobut-2-ene
E-2,3-dichlorobut-2-ene
Z-2,3-dichlorobut-2-ene
Z-1,4-dichlorobut-2-ene
Choose your answer
Was this exam question helpful?
Four atoms, A-D, are labelled in the structure below
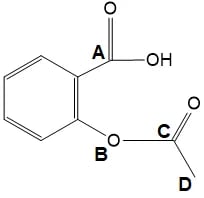
Which atom has a v-shaped arrangement of bonds around it?
Choose your answer
Was this exam question helpful?
Which row has the correct shape, bond angle and explanation for the following molecules?
Molecule | Shape | Bond angle | Explanation | |
|---|---|---|---|---|
A | Ethanoic acid | Pyramidal around the carbonyl carbon | 107o | The carbon atom is sp2 hybridised so the double bond repels more than the single bonds |
B | [Cu(NH3)4(H2O)2]2+ | Octahedral | 90o | The eight bonding pairs will repel each other equally |
C | Cisplatin | Square planar | 109.5o | Minimises the repulsion between the electrons in the d orbitals |
* D* | Sulfur dioxide | Bent | 119o | Lone pairs repel more than bonding pairs due to a higher concentration of the electron charge cloud |
Choose your answer
Was this exam question helpful?
Which statement(s) is/are correct?
Bromoethane is more reactive than chloroethane because bromine is less electronegative than chlorine.
The C-Br bond in bromoethane requires more energy to break than the C-Cl bond in chloroethane due to the C-Br bond being more polar.
The C-Br bond in bromoethane is stronger than the C-Cl bond in chloroethane due to bromine having an increased nuclear charge.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Which statement(s) is/are correct?
Butane and propanone have the same number of electrons so their induced dipole-dipole intermolecular forces are the same.
Only glycine will dissolve in water because it has hydrogen bonding.
Within a protein, glycine has one place in which a hydrogen bond could form.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?