Which pair of compounds will not form a condensation polymer?
A dicarboxylic acid and a diamine
An amino acid and a carboxylic acid
A dicarboxylic acid and a diol
Two amino acids
Was this exam question helpful?
Exam code: H432
Select a download format for Polymers
Select an answer set to view for
Polymers
Which pair of compounds will not form a condensation polymer?
A dicarboxylic acid and a diamine
An amino acid and a carboxylic acid
A dicarboxylic acid and a diol
Two amino acids
Choose your answer
Was this exam question helpful?
The structure below shows the repeating pattern of a polymer.

Which pair of compounds could react rapidly to form this polymer?
Butane-1,4-diol and propanedioic acid
Propane-1,3-diol and butanedioic acid
Butanoyl chloride and propane-1,3-diol
Propane-1,3-diol and butanedioyl dichloride
Choose your answer
Was this exam question helpful?
Which compound can form a condensation polymer by itself?
HOOCCH2CH2COOH
NH2C6H4NH2
COCl CH2NH2
CH2OHCH2COOCH2OH
Choose your answer
Was this exam question helpful?
What are the products of the alkaline hydrolysis of a polyester?
Dicarboxylic acids and compounds with 2 ammonium ion groups
Dicarboxylic acid salts and diamines
Diols and dicarboxylic acids
Diols and dicarboxylic acid salts
Choose your answer
Was this exam question helpful?
The structure below shows the repeating pattern of a polymer.

Which pair of compounds could react to form this polymer?
CH2OHCF(CF3)OH and HOOCCF2CF2COOH
CH2=CFCF3 and C2F4
CH3C(CF3)HF and CF2=CF2
CF3CF=CF2 and CF2HCH3
Choose your answer
Was this exam question helpful?
The repeating unit of a polymer is shown.
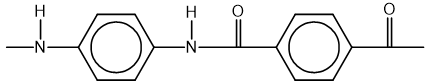
Which monomer or pair of monomers could be used to make this polymer?
Benzene-1,4-dicarboxylic acid and benzene-1,3-diamine
Benzene-1,4-dicarboxylic acid only
Benzene-1,4-dicarboxylic acid and benzene-1,4-diamine
Benzene-1,3-dicarboxylic acid and benzene-1,4-diamine
Choose your answer
Was this exam question helpful?
The repeating unit of a polyester is shown.
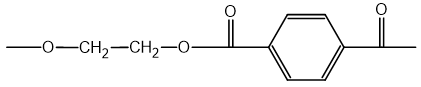
Which statement explains why plastics made from this polyester only soften at high temperatures?
The polymer chains contain strong carbon-carbon bonds
The polymer chains contain hydrogen bonding and London dispersion forces between them
The polymer chains contain permanent dipole-dipole forces and London dispersion forces between them
The polymer chains contain strong carbon-oxygen bonds
Choose your answer
Was this exam question helpful?
Which polymer is most likely to biodegrade in landfill after several years?
PVC
Polyethene
Teflon
Nylon
Choose your answer
Was this exam question helpful?
Part of the Kevlar ® polymer chain is shown.
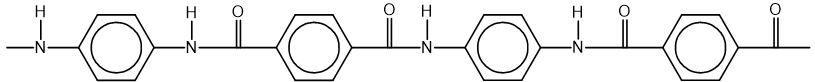
Which monomer or pair of monomers could be used to make Kevlar ®?
Benzoic anhydride and 1,4-diaminobenzene
1,4-benzenedicarbonyl dichloride and 1,4-diaminobenzene
Benzene-1,4-dicarboxylic acid and 4-aminobenzoic acid
4-aminobenzoic acid only
Choose your answer
Was this exam question helpful?
The structure of the polymer PGLA is shown below.
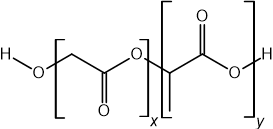
Which compound will be formed from the acid hydrolysis of PLGA?
2-hydroxypropanoic acid
1-hydroxypropanoic acid
Ethanoic acid
Propanoic acid
Choose your answer
Was this exam question helpful?
A section of a condensation polymer is shown below.
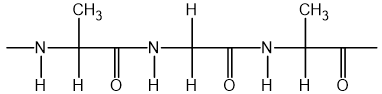
Which monomers react to form this polymer?
3-aminopropanoic acid and aminoethanoic acid
2-aminopropanoic acid and aminoethanoic acid
3-aminobutanoic acid and aminoethanoic acid
Methylamine, ethanoic acid, and propanoic acid
Choose your answer
Was this exam question helpful?
What is the empirical formula of a four-carbon molecule that forms a polymer with ClOC(CH2)8COCl?
C2H6N
C2H3O
C4H9ON
C4H9O2N
Choose your answer
Was this exam question helpful?
Which polymer exhibits the strongest type of intermolecular force?
PET
PVC
Kevlar
Poly(propene)
Choose your answer
Was this exam question helpful?
Polymers can be hydrolysed in acidic or alkaline conditions.
Which row is correct?
| Molecule | Hydrolysis reaction | Product(s) |
|---|---|---|---|
A | Polyester | Alkaline | Diacyl chloride and diol |
B | Polyalkene | Alkaline | Hydroxycarboxylic acid |
C | Polyamide | Acid | Diamine and dicarboxylic acid salt |
D | Protein | Acid | Protonated 2-aminocarboxylic acid |
Choose your answer
Was this exam question helpful?
Which statement about polymers is correct?
Polyesters have higher melting points than polyamides.
The monomer hydroxyethanoic acid has a higher melting point than aminoethanoic acid.
Polyamides are suitable to make fibres for sewing but polyalkenes are not.
Polyalkenes form hydroxycarboxylic acids when hydrolysed in HCl.
Choose your answer
Was this exam question helpful?
The following is a section of the primary structure of a protein.
Gly - Ala - Ser - Leu - Asp
This section was added to 6 mol dm-3 hydrochloric acid and was heated to 110 oC for 24 hours.
How many heterodimeric dipeptides could be formed from the reaction products?
4
10
20
25
Choose your answer
Was this exam question helpful?