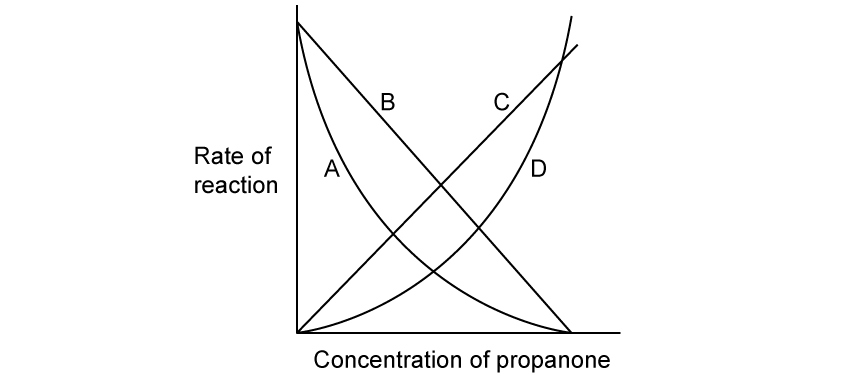
The reaction between iodine and propanone can be catalysed by sulfuric acid. The rate equation shows that this reaction is first order with respect to propanone:
Rate = k [H+] [C3H6O]
Which graph shows that the reaction is first order with respect to propanone?
Was this exam question helpful?