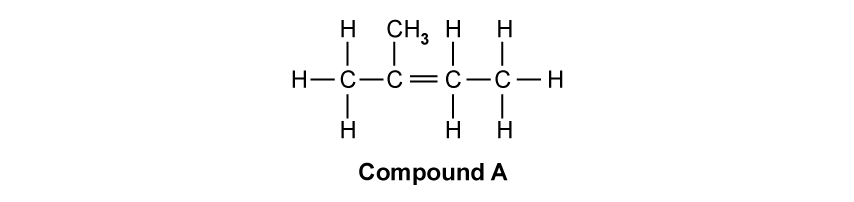
What is the correct systematic name and classification of the alcohol shown?
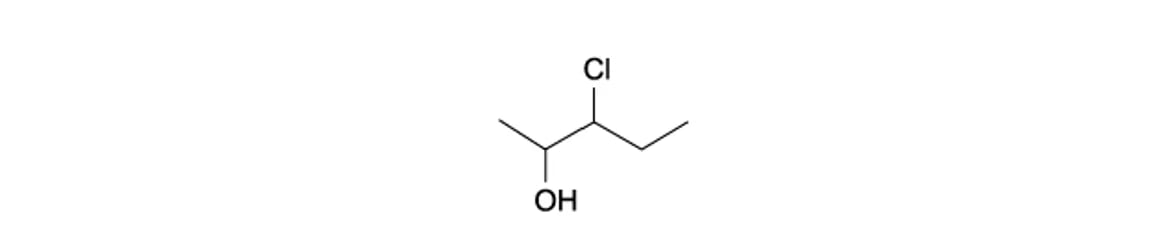
Systematic name | Classification | |
|---|---|---|
A | 2-chloropentan-3-ol | Tertiary |
B | 2-chloropentan-3-ol | Secondary |
C | 3-chloropentan-2-ol | Tertiary |
D | 3-chloropentan-2-ol | Secondary |
Was this exam question helpful?