Nuclear Fusion (OCR A Level Physics): Revision Note
Exam code: H556
Nuclear Fusion
Fusion is defined as:
Small nuclides combine together to make larger nuclei, releasing energy
Low mass nuclei (such as hydrogen and helium) can undergo fusion and release energy
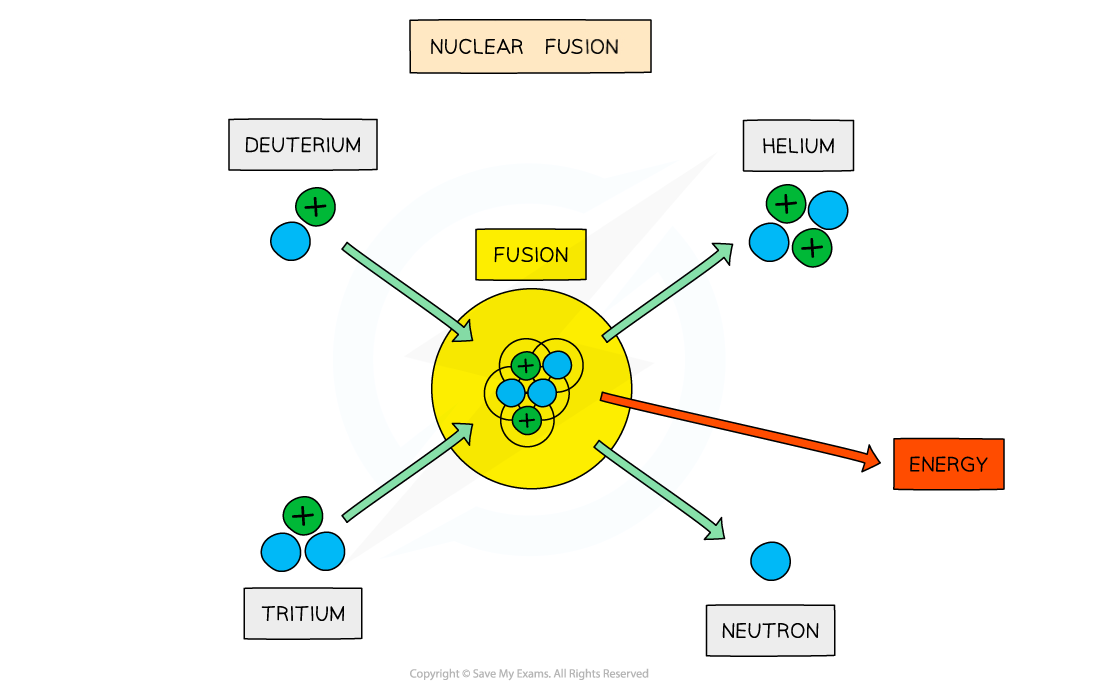
The fusion of deuterium and tritium to form helium with the release of energy
For two nuclei to fuse, both nuclei must have high kinetic energy
This is because the protons inside the nuclei are positively charged, which means that they repel one another
It takes a great deal of energy to overcome the electrostatic force, so this is why it can only be achieved in an extremely high-energy environment, such as a star’s core
When two protons fuse, the element deuterium is produced
In the centre of stars, the deuterium combines with a tritium nucleus to form a helium nucleus, plus the release of energy, which provides fuel for the star to continue burning
Examiner Tips and Tricks
In the fusion process, the mass of the new heavier nucleus is less than the mass of the constituent parts of the nuclei fused together, as some mass is converted into energy.
Not all of this energy is used as binding energy for the new larger nucleus, so energy will be released from this reaction. The binding energy per nucleon afterwards is higher than at the start.

You've read 0 of your 5 free revision notes this week
Unlock more, it's free!
Did this page help you?