Internal Energy of an Ideal Gas (OCR A Level Physics): Revision Note
Exam code: H556
Written by: Katie M
Updated on
Internal Energy of an Ideal Gas
The internal energy of an ideal gas is defined as:
The total kinetic energy of all the particles inside the gas
This is because one of the assumptions of an ideal gas states:
Electrostatic forces between particles in the gas are negligible except during collisions
So, there is no electrostatic potential energy in an ideal gas
Therefore, the internal energy is due to the kinetic energy only
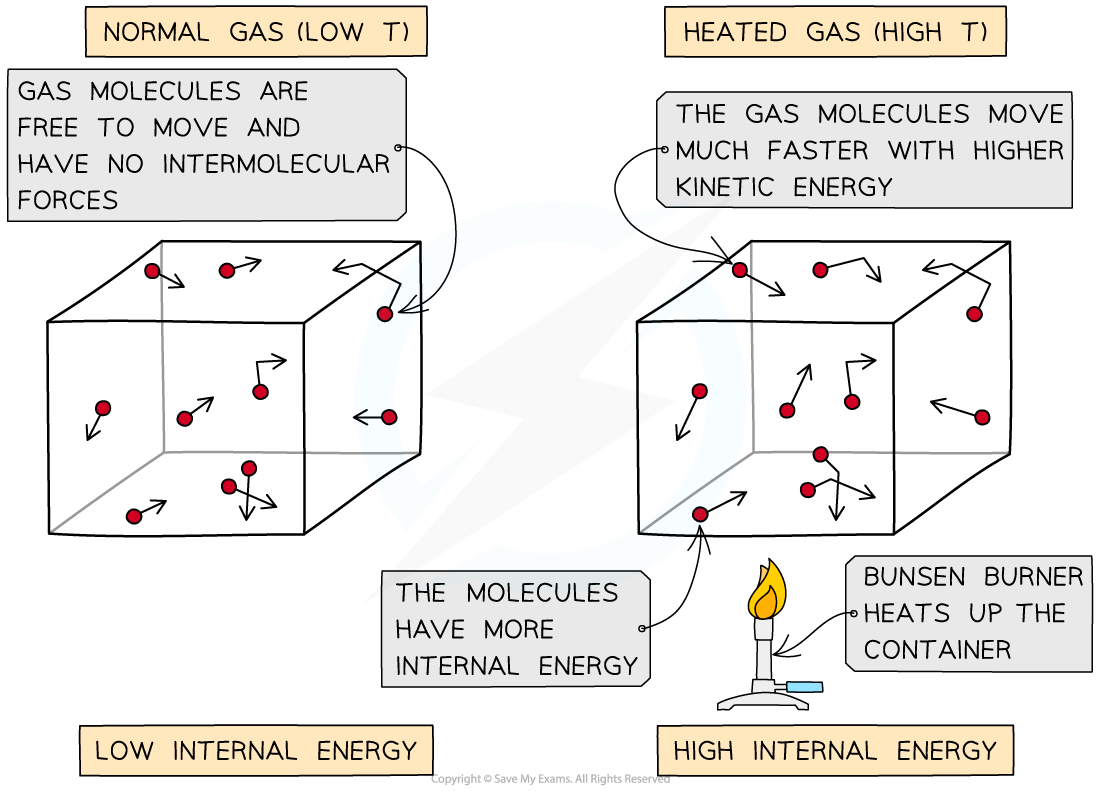
As the container is heated up, the gas molecules move faster with higher kinetic energy and therefore higher internal energy
Change in internal energy, ΔU is equal to the total kinetic energy, EK of all the particles
Where:
EK = total kinetic energy (J)
m = mass of one molecule (kg)
= mean square speed of a molecule (m2 s-2)
k = Boltzmann constant
T = temperature of the gas (K)
N = number of molecules
This equation shows that doubling the temperature will also double the internal energy of the particles
Examiner Tips and Tricks
For a real gas, internal energy is defined as the sum of the kinetic and potential energies of the particles. But for an ideal gas, make sure your definition only refers to kinetic energy. In an exam, you would lose marks for implying there is potential energy (or any other energy) added to the kinetic energy.

Unlock more, it's free!
Was this revision note helpful?
