Specific Heat Capacity (SQA National 5 Physics): Revision Note
Exam code: X857 75
Specific heat capacity
The specific heat capacity,
, of a substance is defined as:
The amount of heat energy required to raise the temperature of 1 kg of the substance by 1 °C per kilogram of mass (J kg-1 °C-1)
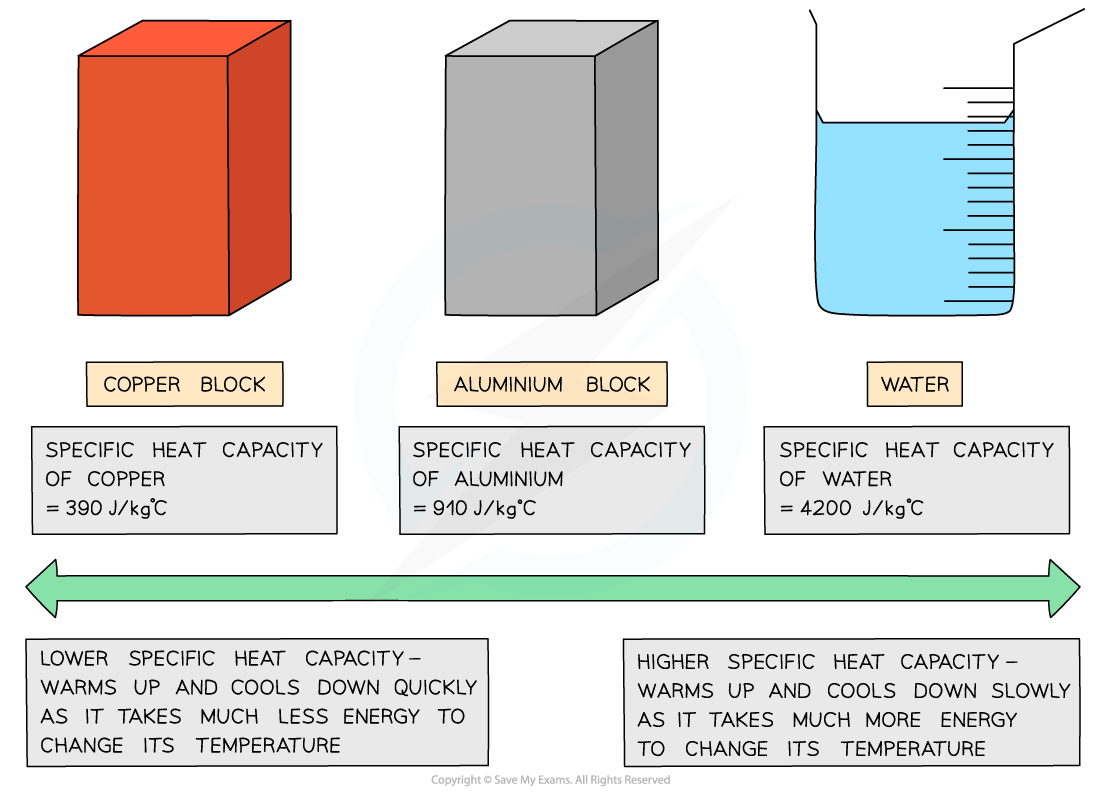
Different substances have different specific heat capacities
If a substance has a low specific heat capacity, it heats up and cools down quickly (ie. it takes less heat energy to change its temperature)
If a substance has a high specific heat capacity, it heats up and cools down slowly (ie. it takes more heat energy to change its temperature)
Examples of specific heat capacity
How much the temperature of a system increases depends on:
The mass of the substance heated
The type of material
The amount of energy put into the system in the form of thermal energy
Worked Example
Two substances of equal mass are both solid at .
An equal amount of heat energy per second is supplied to both substances.
The graph shows the change in temperature over time for each substance.

Identify the substance with the greatest specific heat capacity, and explain your reasoning.
Answer:
A substance with a high specific heat capacity heats up more slowly than a substance with a low specific heat capacity
Substance B heats up more slowly than substance A
Therefore, substance B has a greater specific heat capacity
Examiner Tips and Tricks
Students often lose marks in the exam for not understanding that:
if it takes a lot of energy to change the temperature of a substance, it has a low specific heat capacity
if it takes a small amount of energy to change the temperature of a substance, it has a high specific heat capacity
Make sure you are clear on this and can apply it to new contexts such as graphs and data.

Unlock more, it's free!
Was this revision note helpful?
