Specific Latent Heat (SQA National 5 Physics): Revision Note
Exam code: X857 75
Specific latent heat
A certain amount of energy is required to change the state of a certain mass of a substance
This amount of energy is known as the latent heat
The specific latent heat is defined as:
The amount of energy required to change the state of 1 kg of a substance with no change in temperature
Latent heat is represented by the symbol
with units joules per kilogram (J kg-1)
Change of state
The state or phase of a substance is whether it is a solid, liquid or gas
A change of state, sometimes called a phase change, is when a substance changes from one state to another
For example, when:
a solid turns to a liquid (melting)
a liquid turns to a gas (evaporation)
State changes occur at the melting and boiling point of a substance
Melting and freezing occur at the melting point
Evaporation and condensing occur at the boiling point
Latent heat of fusion & vaporisation
There are two types of specific latent heat:
Specific latent heat of fusion
Changing the state between a solid and liquid
Solid to liquid, or liquid to solid
Specific latent heat of vaporisation
Changing the state between a liquid and gas
Liquid to gas, or gas to liquid
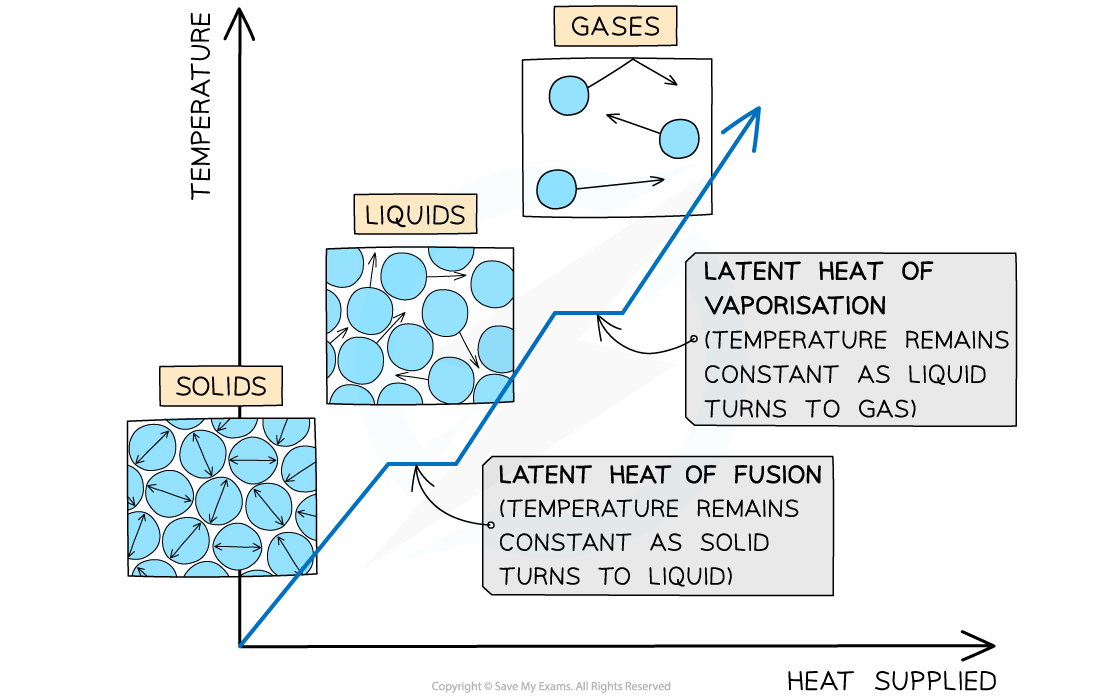
It is important to note that there is no change in temperature whilst a substance is undergoing a change of state
State changes can be identified on a graph by the areas of constant temperature
Latent Heat of Fusion
The specific latent heat of fusion is defined as:
The energy required to convert 1 kg of a substance between a solid and a liquid state with no change in temperature
This applies when melting a solid or freezing a liquid
When a solid substance is melted, its temperature stays constant until all of the substance has melted
The latent heat of fusion is the amount of energy needed per kg for all the particles in the substance to overcome the intermolecular forces of attraction holding them together in their solid state
If a substance in its liquid state is frozen, the substance will solidify at the same temperature as its melting point
In this case, the latent heat of fusion is the amount of energy per kg transferred away from the substance until all the particles in the substance have succumbed to the intermolecular forces of attraction that hold them together in their solid structure
Latent Heat of Vaporisation
The specific latent heat of vaporisation is defined as:
The energy required to convert 1 kg between a liquid and a gaseous state with no change in temperature
This applies when vaporising a liquid or condensing a gas
When a liquid substance is vaporised, its temperature will stay constant until all of the substance has vaporised
The latent heat of vaporisation is the amount of energy per kg needed for all the particles in the substance to overcome the intermolecular forces of attraction holding them together in their liquid state
If a substance in a gas and is condensed, it will condense at the same temperature as its boiling point
In this case, the latent heat of vaporisation is the amount of energy per kg transferred away from the substance until all the particles in the substance have succumbed to the intermolecular forces of attraction that hold them together in their liquid state
Examiner Tips and Tricks
The specific latent heat of fusion and vaporisation value of all substances will be provided for you in the data sheet, so you do not need to memorise any values.
Make sure to include 'with no change in temperature' in your definition of specific latent heat to be awarded full marks.
Use these reminders to help you remember which type of latent heat is being referred to:
Latent heat of fusion = imagine ‘fusing’ the liquid molecules together to become a solid
Latent heat of vaporisation = “water vapour” is steam, so imagine vaporising the liquid molecules into a gas
But remember that the change of state can go in either direction!
Latent just means hidden. The energy being transferred into the substance (by heating it) causes the temperature to rise to the melting point of a substance; we can see this happening using a thermometer. But when the substance reaches its melting point, we can continue to transfer energy to the system, but we don’t see the temperature rising any more; it stays at the melting point. It appears to be hidden or latent because we can’t see its effects.
The period of latent energy transfer happens as the substance is changing state. Therefore, the energy transferred to the substance at this point must be used for the state change.
The energy is transferred to the molecules or particles, and they use it to overcome the intermolecular forces of attraction holding them in their solid or liquid state.

Unlock more, it's free!
Was this revision note helpful?
