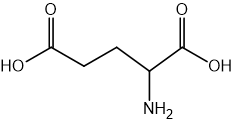
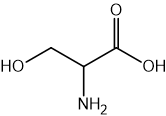
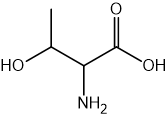
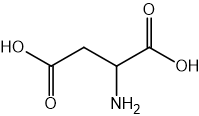
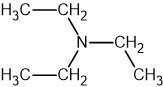
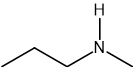
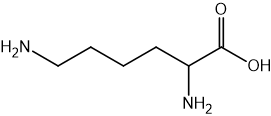
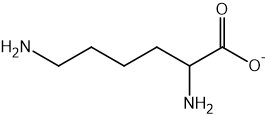
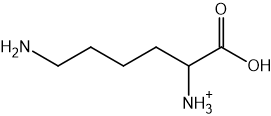
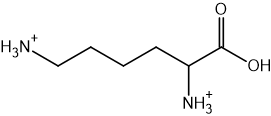
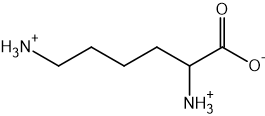
Two reactions are shown below.
Reaction 1: C6H5NO2 → C6H5NH2
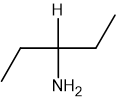
Reaction 2: CH3CH2CHClCH3 → CH3CH2CH(NH2)CH3
Which row shows the correct set of reagents and conditions for each reaction?
| Reaction 1 | Reaction 2 |
|---|---|---|
A | NaBH4 | nitric acid / 50 °C |
B | NaBH4 | nitric acid / <10 °C |
C | Sn / conc. H2SO4 | dilute ethanolic NH3 |
D | Sn / conc. HCl | excess ethanolic NH3 |
Was this exam question helpful?