Combustion of Alkanes (AQA AS Chemistry): Revision Note
Exam code: 7404
Combustion
Alkanes are combusted (burnt) on a large scale for their use as fuels
Complete combustion
When alkanes burn in excess oxygen, complete combustion occurs
All carbon atoms are oxidised to carbon dioxide, and all hydrogen atoms are oxidised to water.
For example, the complete combustion of octane is shown by the balanced equation:

Incomplete combustion
When alkanes burn in limited oxygen, incomplete combustion occurs, and the carbon is not fully oxidised
Instead of forming only carbon dioxide, some carbon is partially oxidised to carbon monoxide
For example, the incomplete combustion of octane, forming carbon monoxide, can be represented by the balanced equation:

Incomplete combustion often takes place inside a car engine due to a limited amount of oxygen present
With a reduced supply of oxygen, carbon will be produced in the form of soot:

The formation of carbon from the incomplete combustion of octane
Combustion & The Environment
Car exhaust fumes contain toxic gases such as carbon monoxide (CO), nitrogen oxides (NO and NO₂, often referred to as NOx), and volatile organic compounds (VOCs)
When released into the atmosphere, these pollutants contribute to environmental problems such as acid rain, photochemical smog, and global warming
They can also harm human health
Carbon monoxide
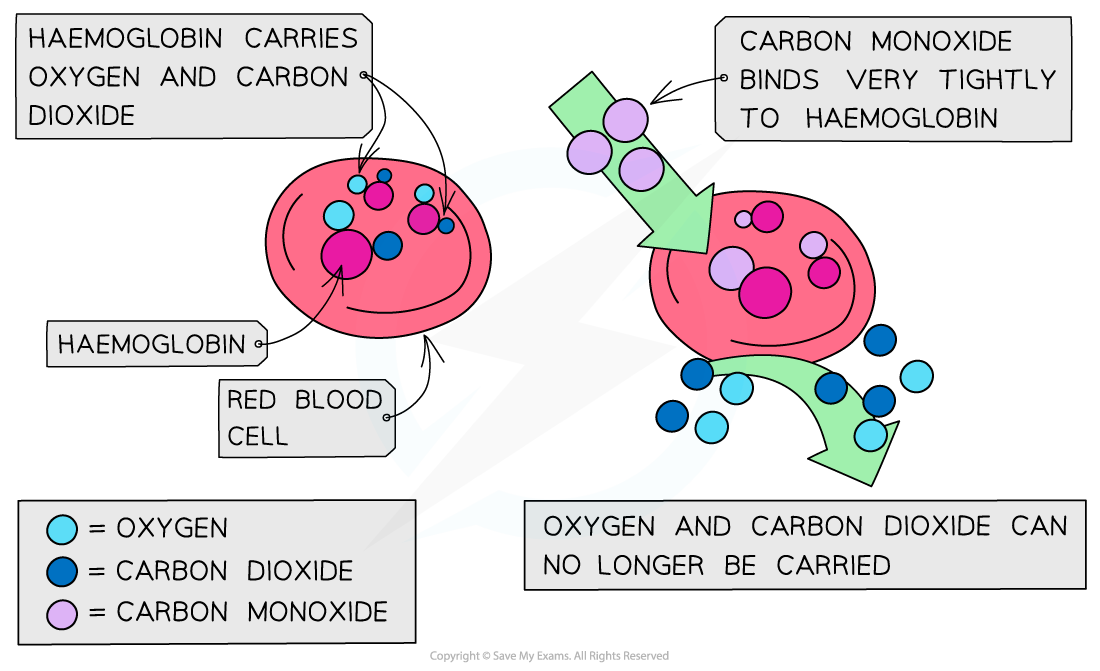
Carbon monoxide (CO) is a toxic, colourless, and odourless gas that can cause dizziness, unconsciousness, and, in severe cases, death
CO binds strongly to haemoglobin in red blood cells, reducing its ability to bind and transport oxygen
Normally, haemoglobin carries oxygen from the lungs to body tissues and transports carbon dioxide from tissues back to the lungs for removal
Oxides of nitrogen
Nitrogen is normally too unreactive to react with oxygen in the air
However, in a car engine, very high temperatures and pressures are reached
Under these conditions, nitrogen reacts with oxygen to form nitrogen monoxide:
N2(g) + O2(g) → 2NO(g)
The nitrogen monoxide can then be further oxidised in air to form nitrogen dioxide:
2NO(g) + O2(g) → 2NO2(g)
Nitrogen oxides can also dissolve and react in water with oxygen to form nitric acid which is a cause of acid rain
Acid rain can cause corrosion of buildings, endangers plant and aquatic life (as lakes and rivers become too acidic) as well as directly damaging human health
These nitrogen oxides (NO and NO2, collectively called NOx) are released into the atmosphere in car exhaust fumes
Car exhaust fumes also contain unburnt hydrocarbons, which are volatile organic compounds (VOCs)
In sunlight, nitrogen oxides react with VOCs to form peroxyacetyl nitrate (PAN), a major component of photochemical smog
PAN can irritate the lungs and eyes, and can also damage plants
Catalytic removal
To reduce the emission of harmful gases, most modern cars are fitted with catalytic converters
Precious metals such as platinum, palladium, and rhodium are coated onto a ceramic honeycomb structure
This provides a large surface area for the catalytic reactions to take place.
The main reactions occurring in a three-way catalytic converter are:
Oxidation of carbon monoxide to carbon dioxide:
2CO + O2 → 2CO2
Reduction of nitrogen monoxide to nitrogen:
2CO + 2NO → 2CO2 + N2
Oxidation of unburnt hydrocarbons to carbon dioxide and water (for an alkane):
CnH2n+2 + (3n+1)[O] → nCO2 + (n+1)H2O
These reactions convert toxic carbon monoxide, nitrogen oxides, and unburnt hydrocarbons into less harmful gases such as carbon dioxide, nitrogen, and water
Reducing sulfur dioxide emissions
Sulfur dioxide emissions from coal-fired power stations can be reduced by treating the waste gases before they are released into the atmosphere
The waste gases are passed through a scrubbing tower, where a slurry containing calcium oxide or calcium carbonate is sprayed into the gases
This process is known as flue gas desulfurisation (FGD), or sulfur scrubbing
When calcium oxide is used, it first reacts with sulfur dioxide and water to form calcium sulfite
The calcium sulfite is then oxidised to form calcium sulfate (gypsum):
CaO (s) + 2H2O (l) + SO2 (g) + ½O2 (g) → CaSO4.2H2O (s)
Calcium carbonate can also be used
It reacts with sulfur dioxide and oxygen to form calcium sulfate and carbon dioxide:
CaCO3 + ½O2 (g) + SO2 (g) → CaSO4 (s) + CO2 (g)
This process removes sulfur dioxide from flue gases and reduces the formation of acid rain
Sulfur dioxide scrubber

Pollutants, their effects, and removal
Pollutant | Formation | Environmental Consequence | Catalytic Removal |
|---|---|---|---|
carbon monoxide | Incomplete combustion of hydrocarbons | Toxic | Oxidation to CO2: 2CO + O2 → 2CO2 and 2CO + 2NO → 2CO2 + N2 |
NOx | Oxidation of nitrogen in a car engine | Dissolve in water with oxygen to form acid rain | Reduction to N2: 2CO + 2NO → 2CO2 + N2 |
VOCs | Unburnt hydrocarbons from fuels | Reaction with NOx in the atmosphere to form PAN | Oxidise unburnt hydrocarbons: CnH2n+2 + (3n+1)[O] → nCO2 + (n+1)H2O |
PAN | From the photochemical reaction of VOCs and nitrogen oxides in the atmosphere | Photochemical smog | Oxidise unburnt hydrocarbons and reduce NOx to prevent the formation of PAN |
Examiner Tips and Tricks
Although carbon dioxide (CO2) is not toxic, it is considered a pollutant because it is a greenhouse gas that contributes to global warming and climate change.

Unlock more, it's free!
Was this revision note helpful?
