Electrophilic Addition (AQA AS Chemistry): Revision Note
Exam code: 7404
Mechanism: Electrophilic Addition
Electrophilic addition
Alkenes undergo electrophilic addition reactions
In an electrophilic addition reaction, two reactants combine to form a single product, giving a 100% atom economy.
Alkenes are particularly reactive because of the C=C double bond, which is an electron-rich region of the molecule
This double bond is readily attacked by positively charged electrophiles.
Alkenes undergo electrophilic addition reactions with hydrogen halides, halogens, and concentrated sulfuric acid (followed by reaction with steam)
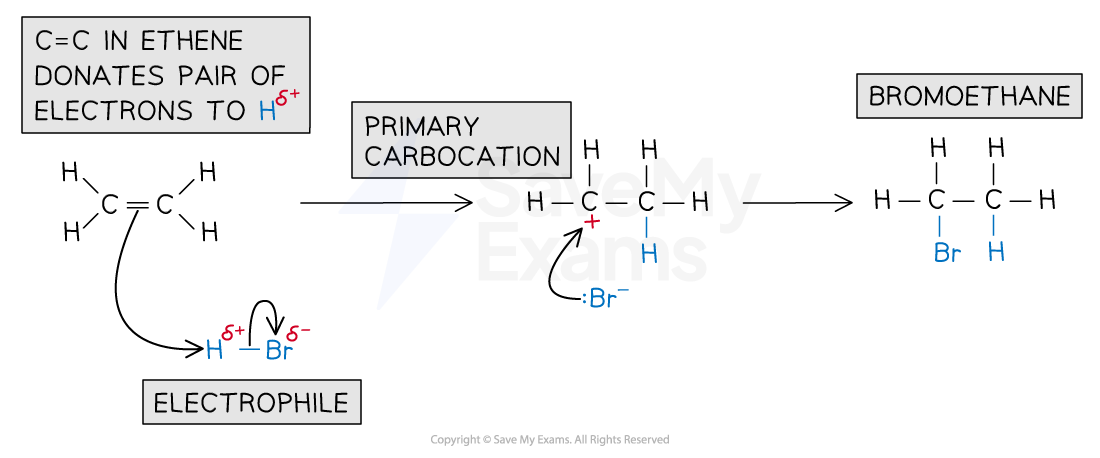
Electrophilic addition of hydrogen bromide
A molecule of hydrogen bromide (HBr) is polar as the hydrogen and bromine atoms have different electronegativities
The bromine atom has a stronger pull on the electrons in the H-Br bond
As a result of this, the Br atom has a partial negative charge and the H atom a partial positive charge

The H atom acts as an electrophile and accepts a pair of electrons from the C-C bond in the alkene
The H-Br bond breaks heterolytically, forming a Br- ion
This results in the formation of a highly reactive carbocation intermediate, which reacts with the bromide ion, Br-
The reaction product is bromoethane:
Electrophilic addition of bromine
Bromine (Br2) is a nonpolar molecule because the two bromine atoms have the same electronegativity and share the bonding electrons equally
However, when a bromine molecule approaches the C=C double bond of an alkene, the high electron density in the double bond repels the electron pair in the Br–Br bond away from the nearer bromine atom
This effect is known as induced polarity
As a result, the bromine atom closest to the double bond becomes slightly positively charged, while the other bromine atom becomes slightly negatively charged

Induced polarity in a bromine molecule During the addition reaction, the bromine atom closest to the alkene acts as an electrophile and accepts a pair of electrons from the C=C bond
The Br–Br bond then breaks heterolytically, forming a Br⁻ ion
This leads to the formation of a highly reactive carbocation intermediate, which is quickly attacked by the Br⁻ ion acting as a nucleophile
The final product is a dihaloalkane, in this case, dibromoethane

Electrophilic addition reaction with sulfuric acid
Water is a weak electrophile, so it does not readily undergo addition reactions with alkenes unless a strong acid is present to act as a catalyst
A suitable strong acid for this reaction is sulfuric acid, H2SO4
In acidic conditions, H3O+ acts as the electrophile
The reaction occurs in two steps:
Step 1:
The π electrons in the C=C bond are attracted to H3O+. Heterolytic fission occurs, forming a carbocation intermediateStep 2:
Water acts as a nucleophile and donates a lone pair of electrons to the positively charged carbon atom, forming a C–O bond. An equilibrium is established between the protonated alcohol and the deprotonated product (the alcohol)
The H3O+ ion is regenerated, so it acts as a catalyst
The reaction product is ethanol

Major and minor products from unsymmetrical alkenes
When the reaction takes place with an asymmetrical alkene, two products are possible, and you can predict which one will be the major product
It depends on the stability of the carbocation formed as the intermediate
The nucleophile will bond to the positive carbon atom of the carbocations
The more stable carbocation produces the major product
The less stable carbocation produces the minor product
The stability of carbon carbocations is as follows:
Tertiary > secondary > primary

First step in the reaction with an unsymmetrical alkene
The π electrons in the C=C bond attack the electrophilic hydrogen:

There are two possible intermediates formed:

The major product formed will be from the intermediate with the more stable carbocation, but some of the minor product from the less stable carbocation intermediate will also form
Formation of major and minor products
The complete mechanism for the addition of HBr to an unsymmetrical alkene is as follows:

In the mechanism above, the secondary halogenoalkane is the major product, because a secondary carbocation is more stable than a primary carbocation

Unlock more, it's free!
Was this revision note helpful?
