What is the mechanism for the reaction of propene with bromine?
Electrophilic addition
Electrophilic substitution
Nucleophilic addition
Nucleophilic substitution
Was this exam question helpful?
Exam code: H032
Select a download format for Alkenes
Select an answer set to view for
Alkenes
What is the mechanism for the reaction of propene with bromine?
Electrophilic addition
Electrophilic substitution
Nucleophilic addition
Nucleophilic substitution
Choose your answer
Was this exam question helpful?
Which of the following compounds will give a positive unsaturation test result with bromine water?
|  |  |  |
A | ✓ | ✓ | ✓ |
B | ✓ | X | ✓ |
C | ✓ | ✓ | X |
D | X | ✓ | ✓ |
Choose your answer
Was this exam question helpful?
Which species will not undergo electrophilic addition with an alkene?
HBr
NaOH
Br2
H2SO4
Choose your answer
Was this exam question helpful?
Which reaction produces a major and a minor organic product?
Ethene + hydrogen bromide
Propene + bromine
But-1-ene + hydrogen bromide
But-1-ene + bromine
Choose your answer
Was this exam question helpful?
Which is the most stable carbocation intermediate?

Choose your answer
Was this exam question helpful?
Which monomer can form the following addition polymer?

Propene
But-1-ene
But-2-ene
Pent-1-ene
Choose your answer
Was this exam question helpful?
Consider the reaction between but-1-ene and hydrogen bromide to form the minor product.
Which carbocation is formed in the electrophilic addition mechanism of this reaction?

Choose your answer
Was this exam question helpful?
What is the bond angle around each carbon atom in ethene?
90°
109.5°
120°
180°
Choose your answer
Was this exam question helpful?
Polyvinyl chloride, PVC, is a common polymer with many everyday uses.

What is the systematic name of PVC?
Poly(chloroethane)
Poly(ethane chloride)
Poly(chloroethene)
Poly(ethene chloride)
Choose your answer
Was this exam question helpful?
Which reagents and conditions are required to form ethanol from ethene?
Hydrogen, heat and nickel catalyst
Steam, heat and nickel catalyst
Hydrogen, heat and acid catalyst
Steam, heat and acid catalyst
Choose your answer
Was this exam question helpful?
Which compound is formed when pent-2-ene reacts with bromine?
1,1-dibromopentane
1,2-dibromopentane
2,2-dibromopentane
2,3-dibromopentane
Choose your answer
Was this exam question helpful?
Which molecule is a Z-isomer?




Choose your answer
Was this exam question helpful?
What is the systematic name of the compound below?

E-2-bromobut-2-ene
Z-2-bromobut-2-ene
E-1,2-dimethyl-1-bromoethene
Z-1,2-dimethyl-1-bromoethene
Choose your answer
Was this exam question helpful?
The displayed formula for a hydrocarbon is shown below.
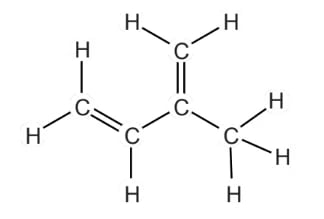
How many σ and π bonds are present in a molecule of this hydrocarbon?
| σ bonds | π bonds |
|---|---|---|
A | 2 | 4 |
B | 10 | 2 |
C | 10 | 4 |
D | 12 | 2 |
Choose your answer
Was this exam question helpful?
Three of the following displayed formulae represent the same isomer of C3H4Cl2 but one structure represents a different isomer, X.
Which displayed formula represents X?
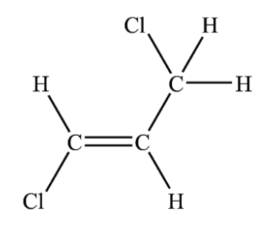



Choose your answer
Was this exam question helpful?
A section of a polymer chain is shown below.

Identify the monomer that would give rise to this section of addition polymer.
E-But-2-ene
Z-But-2-ene
Methylpropene
Propene
Choose your answer
Was this exam question helpful?
A molecule of a polymer contained the sequence shown.

Which monomer could produce this polymer by addition polymerisation?
CH3CCl =CHCl
CH2=CHCl
CHCl =CHCl
CH3CCl =CH2
Choose your answer
Was this exam question helpful?
Isomers X and Y both react with HBr.

A mixture of X and Y is reacted with HBr.
Which three structures represent three different possible products of this reaction?
A | (CH3)2CHCBr3 | (CH3)2CBrCHBr2 | CH3CHBrCHBrCH3 |
B | (CH3)2CHCBr3 | (CH3)2CBrCHBr2 | CH3CBr2CHBrCH3 |
C | (CH3)2CBrCBr3 | (CH3)2CHCBr3 | CH3CBr2CHBrCH3 |
D | (CH3)2CBrCHBr2 | CHBr2CBr(CH3)CH3 | CH3CHBrCBr2CH3 |
Choose your answer
Was this exam question helpful?
Limonene is an oil formed in the peel of citrus fruits.

Which product is formed when an excess of bromine, Br2 (l), reacts with limonene at room temperature in the dark?




Choose your answer
Was this exam question helpful?
Which compound is produced in the reaction between hex-2-ene and steam?
(CH3)2CHCH2CH2CH2OH
CH3CH2CH2CH2CH(OH)CH3
CH3CH2CH(OH)CH2CH3
CH3CH2CH2CH2CH2CH2OH
Choose your answer
Was this exam question helpful?
How many structural isomers of C5H10 react with bromine to form a dibromoalkane?
3
4
5
6
Choose your answer
Was this exam question helpful?
How many σ (sigma) and π (pi) bonds are present in a molecule of phenylacetylene, C6H5CCH?

14 σ and 5 π
14 σ and 3 π
15 σ and 5 π
15 σ and 6 π
Choose your answer
Was this exam question helpful?
The molecule below has two double bonds, labelled 1 and 2.
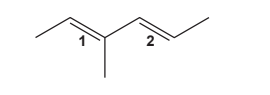
The arrangement around each double bond can be identified as E or Z.
Which row in the table is correct for double bond 1 and double bond 2?
| Double bond 1 | Double bond 2 |
|---|---|---|
A | E | Z |
B | Z | E |
C | E | E |
D | Z | Z |
Choose your answer
Was this exam question helpful?
There are many structural isomers with the molecular formula C4H7Br, including the following:

Of the isomers shown, determine how many
Show E/Z isomerism.
Show optical isomerism.
| Show E/Z isomerism | Show optical isomerism |
|---|---|---|
A | 2 | 1 |
B | 2 | 3 |
C | 3 | 1 |
D | 3 | 3 |
Choose your answer
Was this exam question helpful?
Which statement about propene is correct?
The C-C bond is half as strong as the C=C bond.
It has E/Z isomers due to restricted rotation around the C=C bond.
It undergoes nucleophilic addition with bromine.
It is more reactive than propane due to the strain on the ? bond.
Choose your answer
Was this exam question helpful?
Which molecule does not show E/Z isomerism?
3-methylpent-2-ene
2,4-dimethylpent-2-ene
2,2-dimethylhex-3-ene
1-Propen-1-ylbenzene
Choose your answer
Was this exam question helpful?
Which statement(s) is/are correct about 2-methylbut-1-ene?
In the reaction with steam and H2SO4, the major product formed is 2-methylbutan-2-ol.
During hydrohalogenation, the C=C bond induces a temporary dipole in the hydrogen halide.
The polymer formed can be hydrolysed in the presence of H2SO4.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?