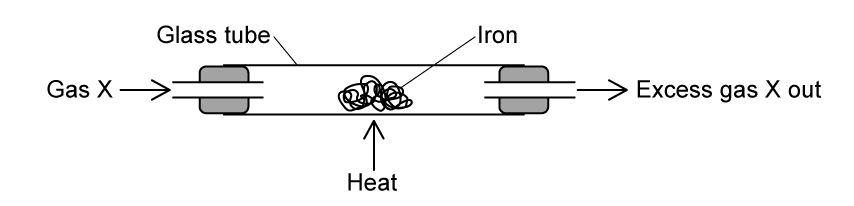The Group 7 elements exist in different states at room temperature.
Draw one straight line from each Group 7 element to its correct state at room temperature.

Chlorine gas will react with sodium to form sodium chloride.
Place a tick (✔) in the box which shows the type of bonding that exists in the compound sodium chloride:
covalent bonding |
|
ionic bonding |
|
metallic bonding |
|
When chlorine reacts with hydrogen, hydrogen chloride is formed.
Complete the dot and cross diagram in Figure 1 for hydrogen chloride.

Figure 1
Use words from the box to complete the sentence about the reactivity of the halogens with hydrogen.
decreases | less | increases | more |
Reactivity ....................................... down Group 7, so iodine reacts ......................................... vigorously with hydrogen than chlorine.
Was this exam question helpful?