Which reaction is an example of nucleophilic substitution?
CH3CH2Br → CH2=CH2 + HBr
CH2=CH2 + HBr → CH3CH2Br
C3H7Br + NH3 → C3H7NH2 + HBr
C2H6 + Br2 → C2H5Br + HBr
Was this exam question helpful?
Exam code: H032
Select a download format for Haloalkanes
Select an answer set to view for
Haloalkanes
Which reaction is an example of nucleophilic substitution?
CH3CH2Br → CH2=CH2 + HBr
CH2=CH2 + HBr → CH3CH2Br
C3H7Br + NH3 → C3H7NH2 + HBr
C2H6 + Br2 → C2H5Br + HBr
Choose your answer
Was this exam question helpful?
What is the correct systematic name and classification of the following molecule?
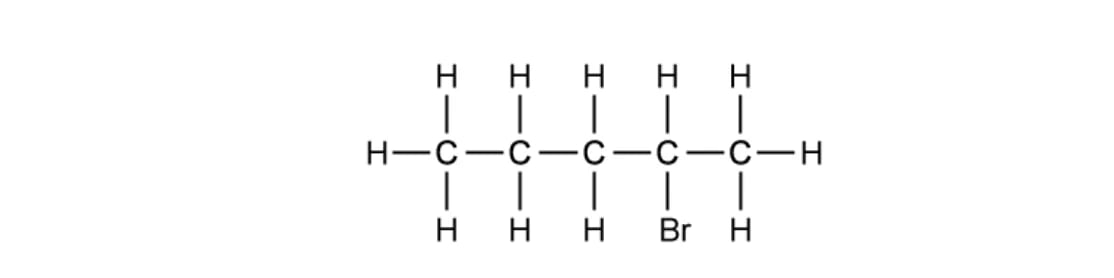
Systematic name | Classification | |
|---|---|---|
A | 1-bromopentane | Primary |
B | 2-bromopentane | Secondary |
C | 2-bromopentane | Primary |
D | 4-bromopentane | Secondary |
Choose your answer
Was this exam question helpful?
Which of the following statements is not correct about the depletion of the ozone layer?
CFCs produce free radicals when they are exposed to UV light in the upper atmosphere
2 moles of ozone break down to form 3 moles of oxygen
Fluorine and chlorine free radicals can break down ozone molecules in the upper atmosphere
The mechanism is a radical-catalysed decomposition reaction
Choose your answer
Was this exam question helpful?
Three haloalkanes are heated separately with aqueous silver nitrate in ethanol. Each reaction produces a precipitate.
Which haloalkane produces a precipitate the fastest?
1-iodopropane
They precipitate at the same rate
1-chloropropane
1-bromopropane
Choose your answer
Was this exam question helpful?
Which statement about a nucleophile is correct?
It is an electron pair donor
It is an electron pair acceptor
It is a proton acceptor
It is a proton donor
Choose your answer
Was this exam question helpful?
Which compound has the fastest rate of reaction with aqueous potassium hydroxide to form an alcohol?
1- chlorobutane
1-bromobutane
1-fluorobutane
1-iodobutane
Choose your answer
Was this exam question helpful?
Which statement about ozone is not correct?
It decomposes to form oxygen
Ozone holes are regions of the upper atmosphere where there is a reduced concentration of ozone
Hydrofluorocarbons are more damaging to the ozone layer than chlorofluorocarbons
It absorbs ultraviolet radiation from the Sun
Choose your answer
Was this exam question helpful?
The equation for the alkaline hydrolysis of 1-iodopropane is shown below
CH3CH2CH2I + OH– →CH3CH2CH2OH + I –
What is the name of the mechanism used for this reaction?
Electrophilic addition
Elimination
Nucleophilic substitution
Oxidation
Choose your answer
Was this exam question helpful?
Which row shows the correct shape and bond angle in a molecule of CF2Cl2?
Shape | Bond angle | |
|---|---|---|
A | Linear | 180° |
B | Pyramidal | 107° |
C | Tetrahedral | 109.5° |
D | Trigonal planar | 120° |
Choose your answer
Was this exam question helpful?
Which statement explains why haloalkanes are more reactive than alkanes?
Carbon is more electronegative than the halogens
The carbon and halogen atoms have different electronegativities
Carbon-halogen bonds are stronger than carbon-hydrogen bonds
The carbon-halogen bond is non-polar
Choose your answer
Was this exam question helpful?
A chemist adds four drops of 1-chloropentane, 1-bromopentane and 1-iodopentane separately to three test-tubes containing 1.0 cm3 of aqueous silver nitrate at 60 °C. A hydrolysis reaction occurred.
Which statement best explains why 1-iodopentane precipitates first and 1-chloropentane precipitates last?
The bond polarity decreases from C-Cl to C-I
The electronegativity of the halogens decreases down the group
The bond enthalpy decreases from C-Cl to C-I
The ionisation energy of the halogens decreases from Cl to I
Choose your answer
Was this exam question helpful?
Which statement best describes the mechanism of the reaction between aqueous sodium hydroxide and 2-bromopropane?
Attack by a nucleophile on a carbocation
Attack by a nucleophile on a carbon atom with a partial positive charge
Homolytic bond fission and attack by a nucleophile on a carbocation
Homolytic bond fission and attack by an electrophile on a partial positive charge
Choose your answer
Was this exam question helpful?
In the hydrolysis of bromoethane by aqueous sodium hydroxide, what is the nature of the attacking species and of the leaving group?
Attacking group | Leaving group | |
|---|---|---|
A | nucleophile | nucleophile |
B | electrophile | nucleophile |
C | nucleophile | electrophile |
D | electrophile | electrophile |
Choose your answer
Was this exam question helpful?
Which reagents and conditions are required for the following reaction?
CH3CH2CH2I → CH3CH2CH2OH
Heated under reflux, aqueous potassium cyanide
Heated under reflux, ethanol, potassium cyanide
Heated under reflux with aqueous sodium hydroxide
Warm with a solution of ethanol
Choose your answer
Was this exam question helpful?
Which pair of reactions shows the correct sequence of attack of CFC molecules with ozone?
| Reaction 1 | Reaction 2 |
|---|---|---|
A | F● + O3 → FO● + O2 | FO● + O3 → 2O2 + F● |
B | Cl + O3 → ClO● + O2 | ClO● + O3 → 2O2 + Cl |
C | Cl● + O3 → ClO● + O2 | ClO● + O3 → 2O2 + Cl● |
D | Cl● + O3 → ClO + O2 | ClO + O3 → 2O2 + Cl● |
Choose your answer
Was this exam question helpful?
What is the correct systematic name and classification of the following molecule?
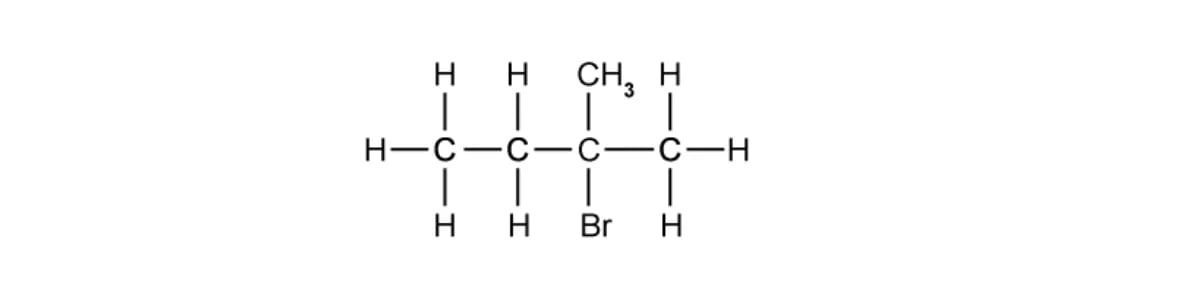
Systematic name | Classification | |
|---|---|---|
A | 2-methyl-2-bromobutane | Tertiary |
B | 3-methyl-3-bromobutane | Secondary |
C | 2-bromo-2-methylbutane | Secondary |
D | 2-bromo-2-methylbutane | Tertiary |
Choose your answer
Was this exam question helpful?
The presence of a halogen in an organic compound may be detected by warming the organic compound with aqueous silver nitrate in ethanol.
Which compound would be the quickest to produce a precipitate?
1,2,3-trichlorobutane
1,2-dichloro-3-fluorobutane
1,2-difluoro-3-iodobutane
1-bromo-2-chloro-3-fluorobutane
Choose your answer
Was this exam question helpful?
The breakdown of the ozone layer can be catalysed by NO● radicals.
Which equation is a propagation step in the mechanism for this process?
NO2● + O → NO● + O
NO● + O3 → NO2● + O2
O3 + O → 2O2
NO● + O2 → NO2● + O3
Choose your answer
Was this exam question helpful?
Dichlorodifluoromethane, CCl2F2, has been used in aerosol propellants and as a refrigerant.
Which statement helps to explain why dichlorodifluoromethane is chemically inert?
The carbon-fluorine bond energy is large
The carbon-fluorine bond has a low polarity
Fluorine is highly electronegative
Fluorine compounds are non-flammable
Choose your answer
Was this exam question helpful?
Which compound does not react with nucleophiles?
CH3CH2CH2OH
CH2CH2CHO
CH3CH2CH2Cl
CH3CH2CH2CH3
Choose your answer
Was this exam question helpful?
Hydrogen bromide reacts with 3-methylbut-1-ene.
What is the structure of the major intermediate formed in the mechanism?




Choose your answer
Was this exam question helpful?
Which statements is/are correct about haloalkanes?
Published bond enthalpies may differ because they are calculated as a mean average across different chemical environments, not just within one compound.
Haloalkanes that contain a C-F bond are the least reactive because this bond has the highest bond enthalpy.
When heated with aqueous silver nitrate in ethanol, the haloalkane that takes the longest to form a precipitate is a chloroalkane.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Which statements is/are correct?
The OH- ion is a better nucleophile than water.
During nucleophilic substitution, the carbon-halogen bond breaks homolytically forming a halide ion.
Nucleophiles are 'nucleus loving' so are themselves positively charged.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
Which statement about haloalkanes is correct?
Haloalkanes and alcohols both take part in dehydration reactions to produce alkenes
Haloalkanes are soluble in water due to the polar carbon-halogen bond
The OH- ion acts as a base in an elimination reaction
Hot ethanolic NaOH is used to produce the nucleophile in a substitution reaction
Choose your answer
Was this exam question helpful?
Which statements is/are not correct about the depletion of the ozone layer?
CH3F will produce a fluorine radical and a CH3 radical in the presence of UV radiation.
CH2ClF is a common example of a hydrofluorocarbon used in refrigerators.
Chlorofluorocarbons are preferred to hydrofluorocarbons because they cause less depletion of the ozone layer.
1, 2 and 3
Only 1 and 2
Only 2 and 3
Only 1
Choose your answer
Was this exam question helpful?
The breakdown of ozone is catalysed by NO radicals.
Which equation shows a propagation step in the mechanism for this process?
NO● + ●NO2 → N2O + O2
NO2● + O → NO● + O2
N2 + O2 → NO● + NO●
NO● + O3 → NO2 + ●O2
Choose your answer
Was this exam question helpful?