Define specific heat capacity.
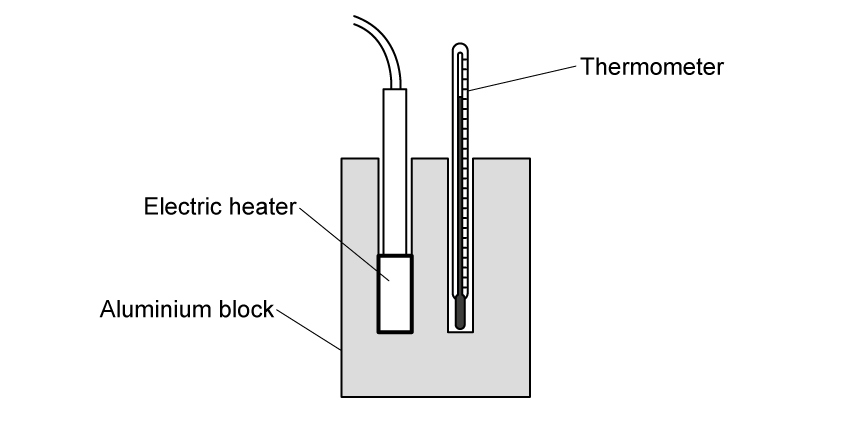
Figure 1 shows some equipment used to determine the specific heat capacity of aluminium.

Figure 1
Complete the missing labels using words or phrases from the box below. Each option is used once.
Electric heater | Insulating container | Thermometer |
Figure 2 shows the reading before the heater was switched on. Figure 3 shows the reading at the end of the investigation.

Figure 2

Figure 3
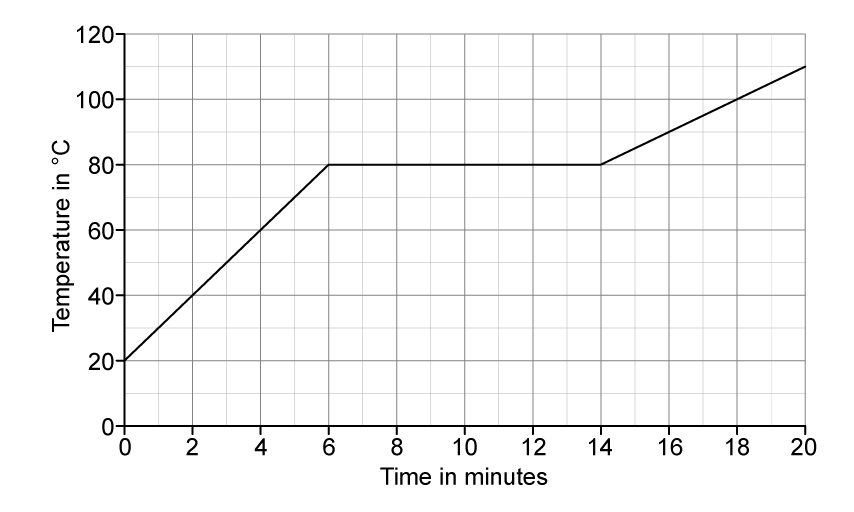
Calculate the change in temperature.
change in temperature = ....................................°C
Calculate the specific heat capacity of aluminium.
The following information may be useful:
Mass of block = 0.850 kg
Energy supplied = 17 000 J
State the correct unit.
specific heat capacity = ................................ unit.............
Was this exam question helpful?