Key Takeaways
Biological catalysts are molecules that speed up chemical reactions in living organisms without being altered themselves
Biological catalysts are called enzymes, which are proteins produced by living cells
Enzymes work by binding substrates to their active sites, forming an enzyme-substrate complex
Temperature, pH, and substrate concentration all affect how quickly biological catalysts work
If conditions become too extreme, enzymes denature and permanently lose their shape
What Are Biological Catalysts?
A catalyst is a substance that speeds up a chemical reaction without being changed or used up in the process. Biological catalysts are called enzymes. Enzymes are proteins made by living cells, and they control virtually every chemical reaction in the cell.
Every living organism, from bacteria to blue whales, relies on enzymes. They're involved in everything from breaking down food to copying DNA. Without them, most reactions in your body would happen far too slowly to sustain life. Digesting a single meal, for instance, would take two to three weeks instead of roughly four hours.
Why Are Enzymes Called Biological Catalysts?
Enzymes earn the label "biological catalyst" for two reasons. First, they behave like any catalyst: they lower the activation energy needed for a reaction to happen, which speeds it up. They also remain unchanged at the end of the reaction, ready to work again immediately.
Second, they're biological. Unlike industrial catalysts such as iron or manganese dioxide, enzymes are produced by living cells. They're made from chains of amino acids folded into precise three-dimensional shapes. That shape is what makes them work.
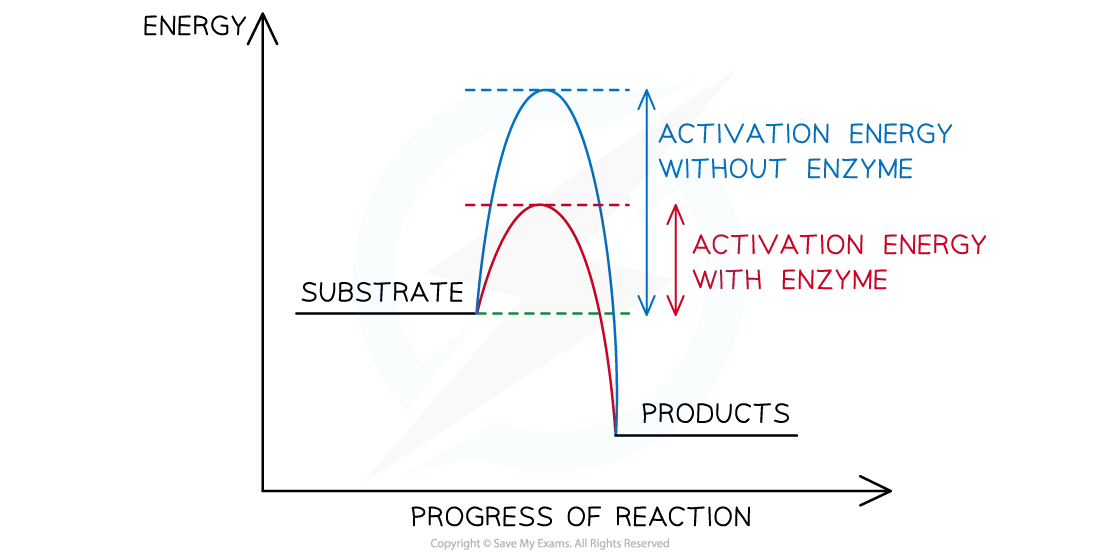
How Do Biological Catalysts Work?
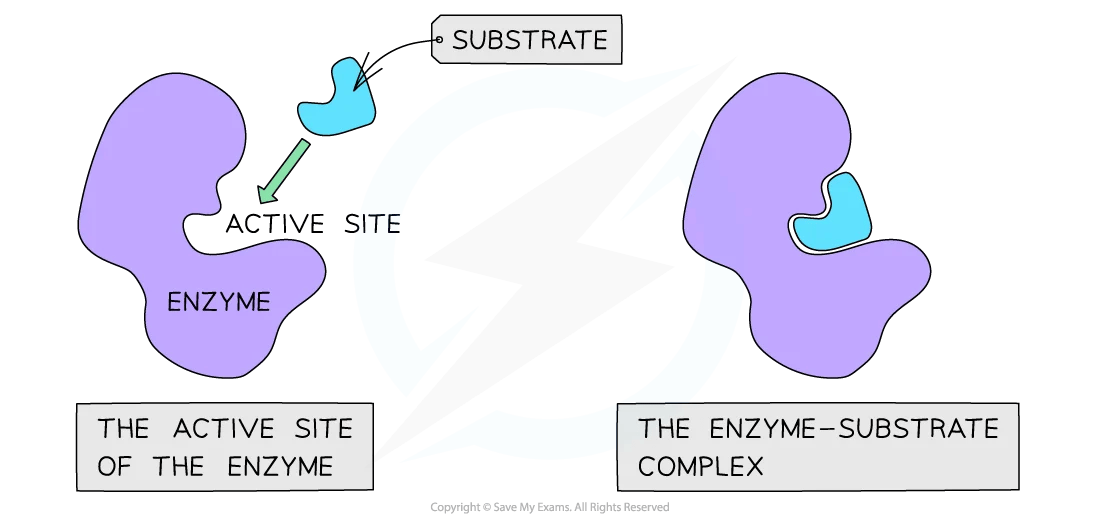
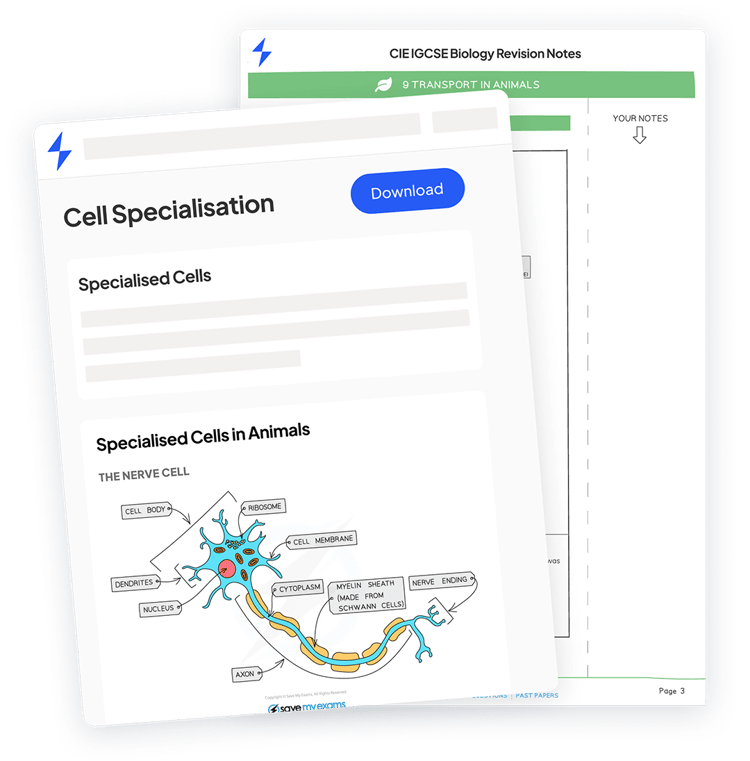
Each enzyme has a region called an active site. This is a small pocket with a very specific shape, and only the right substrate (the molecule the enzyme acts on) can fit into it.
The process works like this:
The enzyme and substrate collide in solution due to random movement
The substrate fits into the active site, forming an enzyme-substrate complex
The reaction takes place, and the products are released
The enzyme is free to accept another substrate
This is often explained using the lock and key model. The enzyme is the lock, and the substrate is the key. They are complementary to one another.
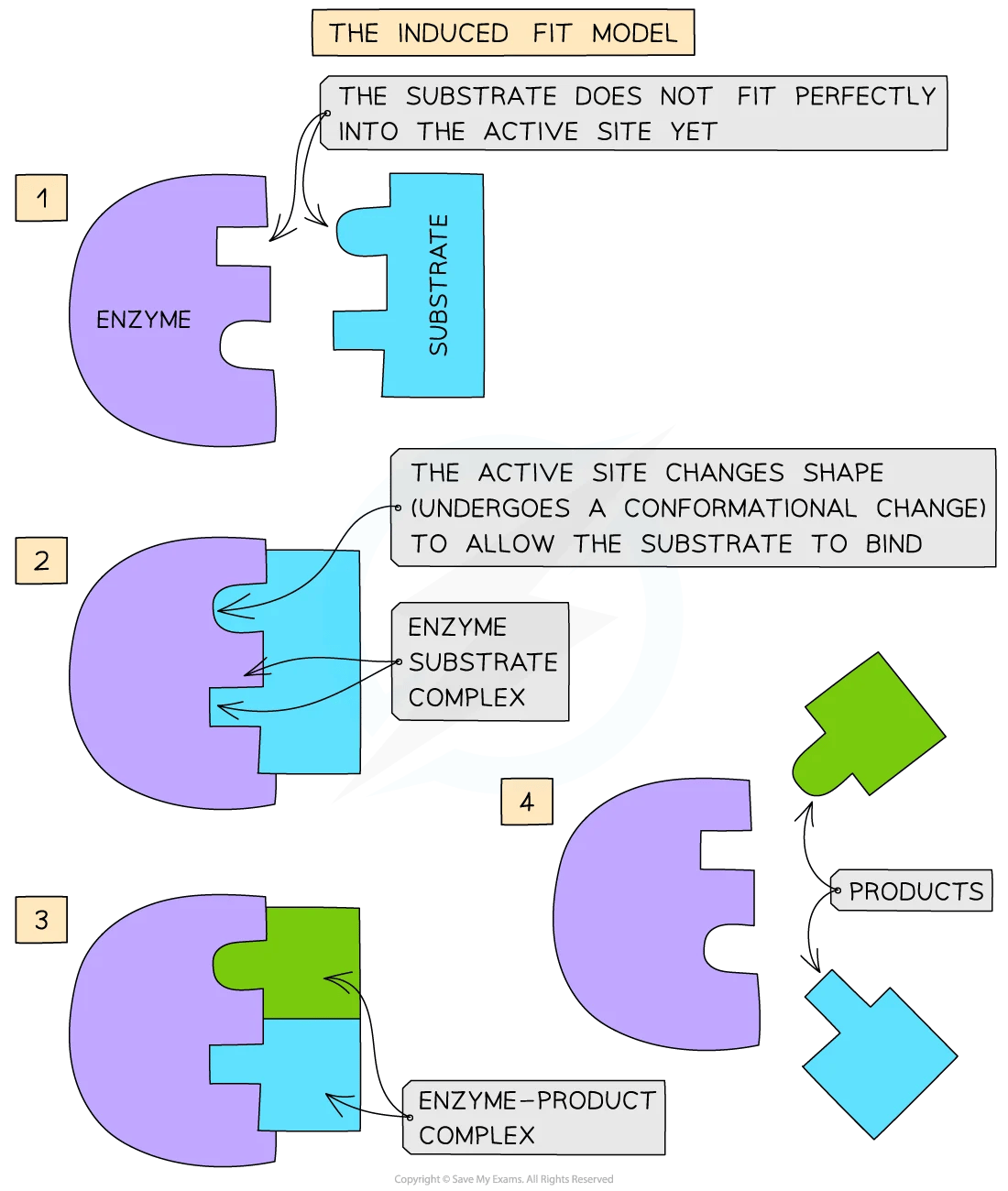
The induced fit model is more sophisticated. It describes the active site as changing shape slightly as the substrate binds, gripping it more tightly.
“Remember: it’s complementary (the enzyme active site and substrate fit together), not complimentary (they don’t say nice things to each other)!”
– Natalie Lawrence, Biology Tutor.
Factors Affecting Biological Catalyst Activity
Enzymes are sensitive to their environment. Three main factors control how fast they work.
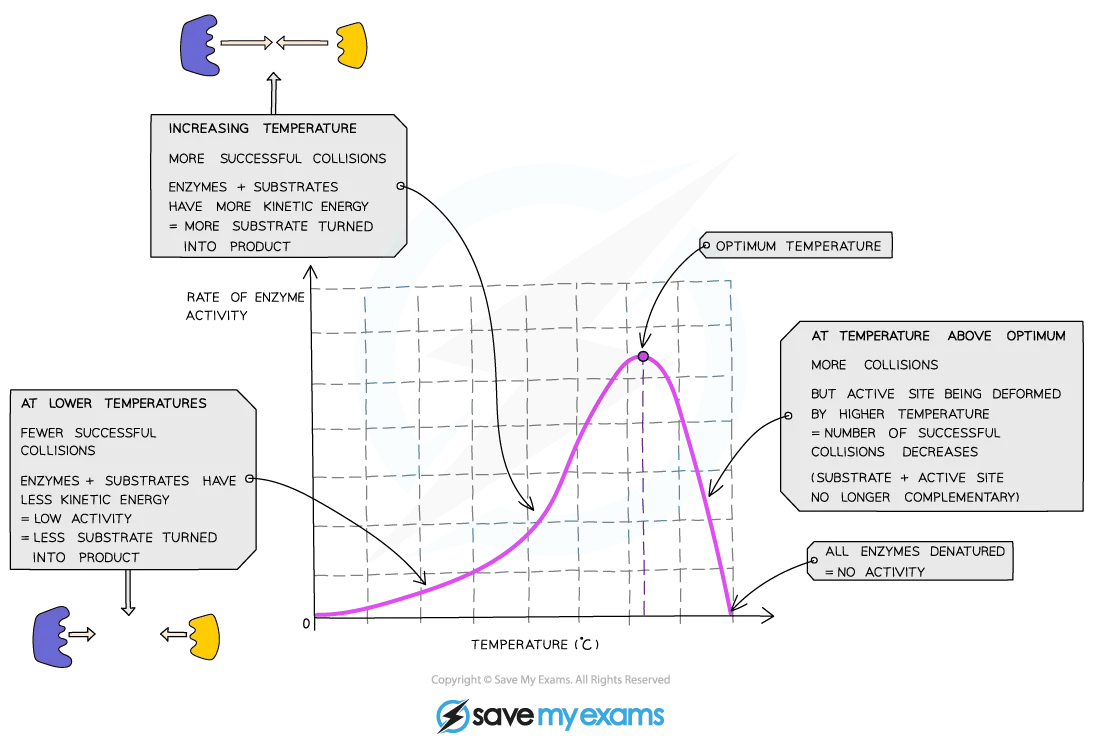
Temperature plays a big role. As temperature rises, molecules move faster and collide more often, so reaction rate increases. Every enzyme has an optimum temperature where it works best. In the human body, that's around 37°C. Go much higher, and the bonds maintaining the enzyme's three-dimensional shape start to break. The active site distorts, the substrate can't fit, and the enzyme is denatured. Low temperatures don't destroy enzymes, but they do slow them down.
pH matters too. Most human enzymes work best at around pH 7, but there are exceptions. Pepsin, which works in the stomach, has an optimum pH of about 2. Enzymes in the small intestine prefer pH 8 or 9. Extremes of pH cause denaturation, just like extreme heat.
Substrate concentration also affects the rate. More substrate molecules mean more collisions with enzymes. But once every active site is occupied, adding more substrate won't speed things up. The enzyme’s active sites are saturated.
Examples of Biological Catalysts
There are thousands of different enzymes, each catalysing a specific reaction. Here are some you'll come across frequently:
Enzyme | Where it works | What it breaks down | Products |
|---|---|---|---|
Amylase | Saliva and small intestine | Starch | Maltose |
Protease | Stomach and small intestine | Proteins | Amino acids |
Lipase | Small intestine | Lipids (fats) | Glycerol and fatty acids |
Catalase | In body cells | Hydrogen peroxide | Water and oxygen |
Enzymes don't just break things down. They also build molecules up. Glycogen synthase, for example, joins glucose molecules together to form glycogen for energy storage.
Biological Catalysts vs Chemical Catalysts
Biological and chemical catalysts both speed up reactions, but they work in very different ways.
Feature | Biological catalysts (enzymes) | Chemical catalysts |
|---|---|---|
Made from | Proteins (amino acids) | Metals or inorganic compounds |
Specificity | Highly specific to one substrate | Often work on multiple reactions |
Working temperature | Body temperature (around 37°C) | Often require extreme heat |
pH sensitivity | Sensitive; denature at extremes | Less sensitive to pH |
Origin | Produced by living cells | Manufactured or mined |
Iron is used as a catalyst in the Haber process to make ammonia, but it needs temperatures above 400°C. Enzymes in your body achieve similar reaction speeds at just 37°C. That efficiency is one reason why biological catalysts are now used in industries like food production and medicine, from making cheese to producing biofuels.
If you're looking for a deeper understanding of how enzymes work in living systems, Save My Exams' AQA GCSE revision notes on Enzymes & Metabolism cover everything from the lock and key model to the effects of temperature and pH, written by experienced teachers and examiners. We also have notes tailored to your specific course.
Frequently Asked Questions
What happens to biological catalysts when they are denatured?
When an enzyme is denatured, the bonds holding its shape break, and the active site changes shape permanently. The substrate can no longer fit, so the reaction stops. Denaturation is caused by extreme temperatures or pH values and can't be reversed.
Can biological catalysts be reused?
Yes. Enzymes are not used up or changed during a reaction. Once the products leave the active site, the enzyme is free to bind with another substrate molecule. A single enzyme can process hundreds or even thousands of substrates per second.
What is the difference between an enzyme and a catalyst?
All enzymes are catalysts, but not all catalysts are enzymes. A catalyst is any substance that speeds up a reaction without being used up in the process. Enzymes are a specific type of catalyst that are biological in origin, made of protein, and highly specific to particular substrates.
Why do biological catalysts only work on specific substrates?
Each enzyme has an active site with a unique three-dimensional shape. Only substrates with a complementary shape can bind to it. This is why one enzyme typically catalyses one reaction. If the shapes don't match, no enzyme-substrate complex forms and no reaction occurs.
Examiner-written GCSE Biology revision resources that improve your grades 2x
- Written by expert teachers and examiners
- Aligned to exam specifications
- Everything you need to know, and nothing you don’t
Was this glossary entry helpful?
Share this article
 written revision resources that improve your
written revision resources that improve your