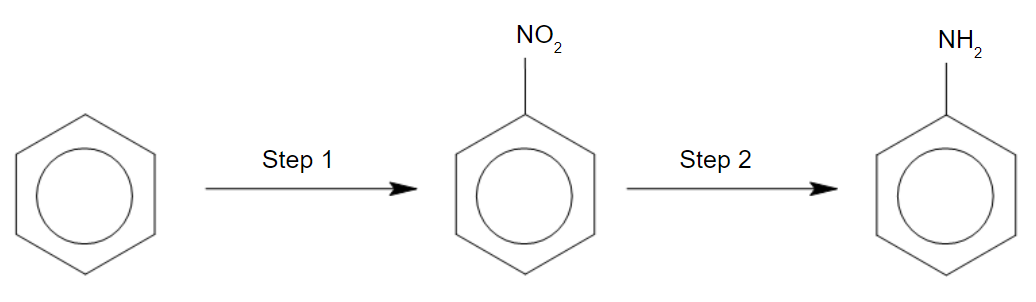
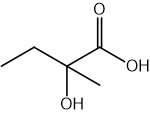
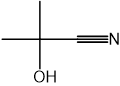
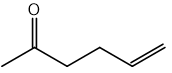
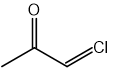
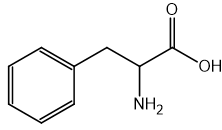
CN– ions react with haloalkanes and with carbonyl compounds.
Which row gives the correct mechanisms for the reactions?
| Reaction of CN- with haloalkanes | Reaction of CN- with carbonyl compounds |
|---|---|---|
A | Electrophilic substitution | Electrophilic addition |
B | Electrophilic substitution | Nucleophilic addition |
C | Nucleophilic substitution | Electrophilic addition |
D | Nucleophilic substitution | Nucleophilic addition |
Was this exam question helpful?