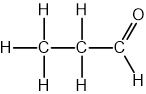
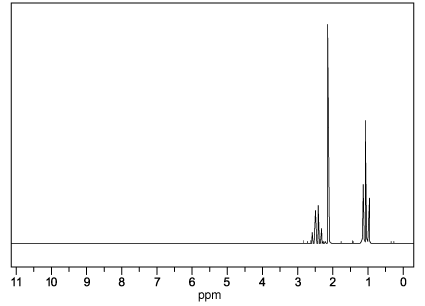
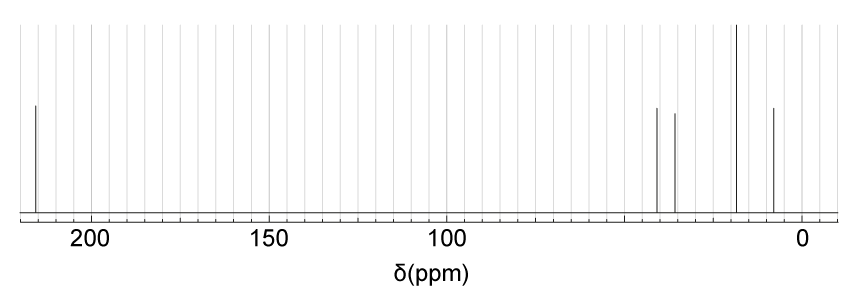
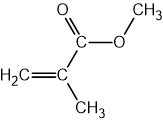
The structure of 2-chloropropane is shown below.
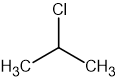
Which description of the peaks in the 1H NMR spectrum of 2-chloropropane is correct?
1 peak with a relative intensity of 6
2 peaks with relative intensities of 3 : 4
3 peaks with relative intensities of 3 : 1 : 3
2 peaks with relative intensities 6 : 1
Was this exam question helpful?