Energy From Fuels (SQA National 5 Chemistry): Revision Note
Exam code: X813 75
Exothermic & Endothermic Reactions
All chemical reactions involve energy changes
These can usually be detected as a change in temperature
There are two main types of energy change:
Exothermic
Endothermic
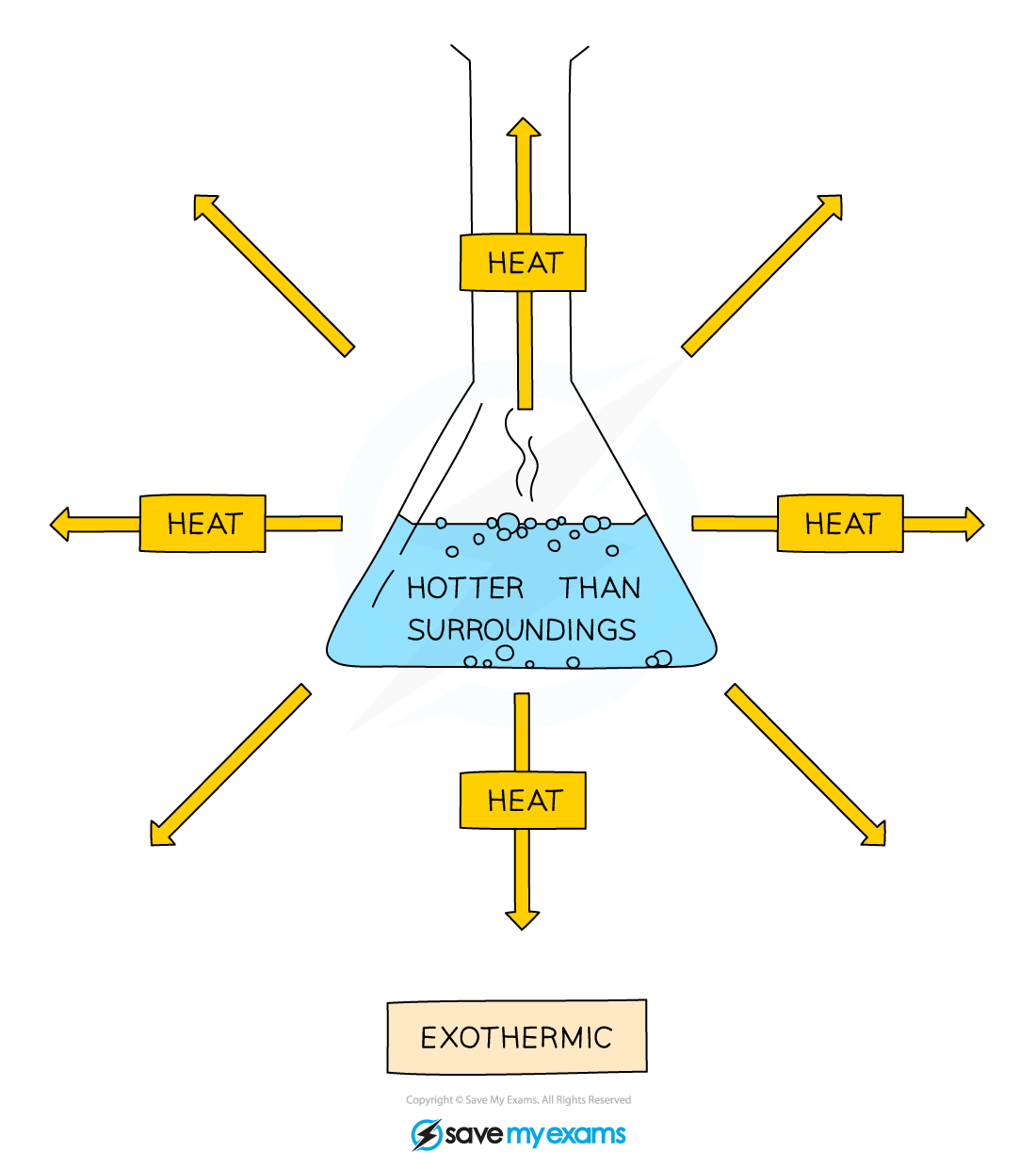
Exothermic reactions
An exothermic reaction releases heat energy into the surroundings
This means that the temperature increases
Neutralisation and combustion are common examples of exothermic reactions
Exothermic reaction diagram
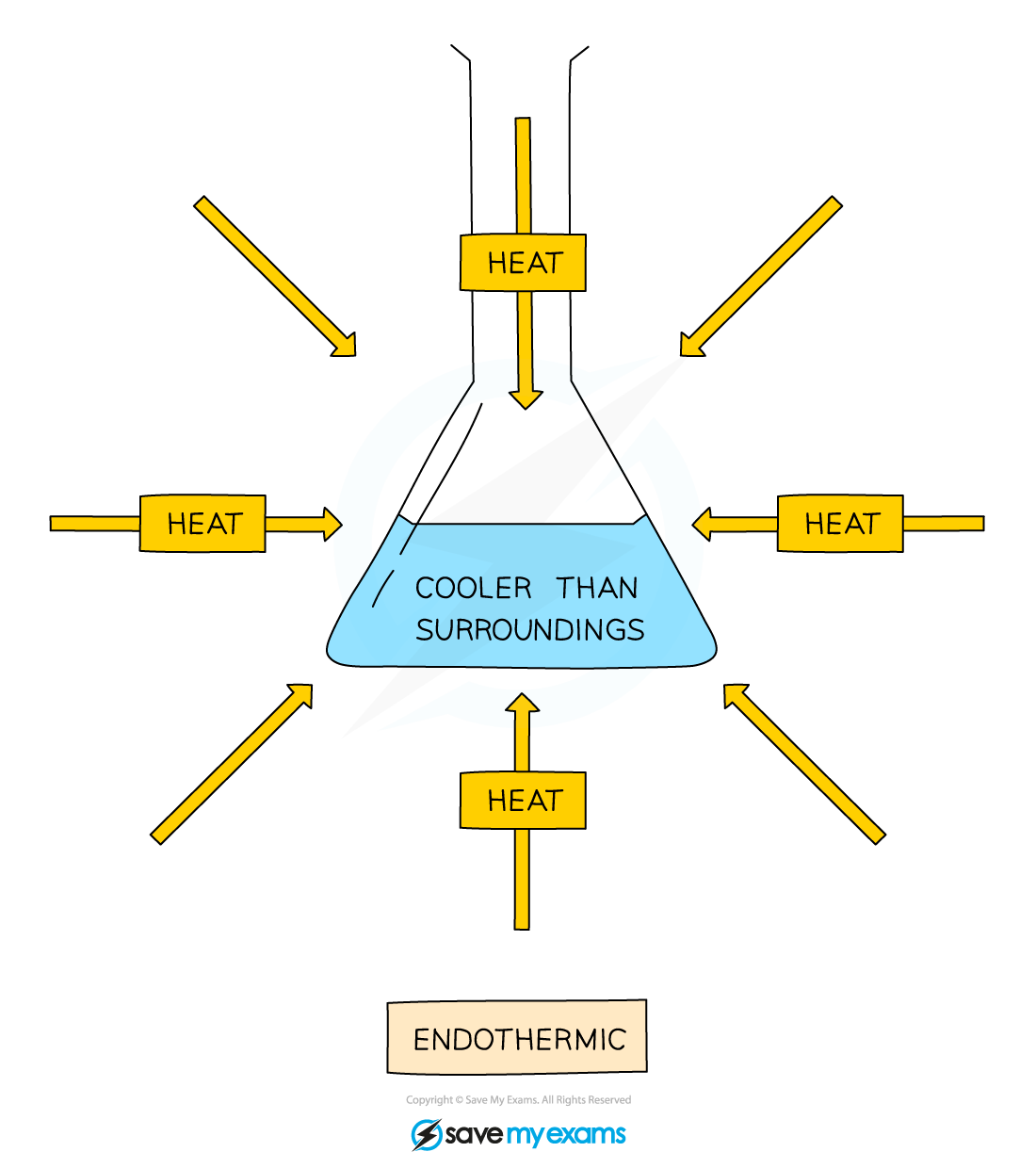
Endothermic reactions
An endothermic reaction takes in heat energy from the surroundings
This means that the temperature decreases
Endothermic reaction diagram
Worked Example
A student investigated four reactions and recorded the temperature change for each. Identify which reactions are exothermic and which are endothermic, giving a reason for your answer.
[2]
Experiment | Initial temperature (oC) | Final temperature (oC) |
1 | 19 | 21 |
2 | 20 | 16 |
3 | 20 | 26 |
4 | 19 | 31 |
Answer:
Reactions 1, 3 and 4 are exothermic
Reaction 2 is endothermic
[1 mark]
The exothermic reactions all show a temperature increase, which means that heat energy has been released
The endothermic reactions all show a temperature decrease, which means that heat energy was taken in
[1 mark]
Examiner Tips and Tricks
You need to able to work with experimental data to identify exothermic and endothermic reactions and justify your decisions
Combustion reactions
Combustion (or burning) is a chemical reaction where a substance reacts with oxygen to release energy
Due to the release of energy, combustion reactions are exothermic
Substances that we burn to release useful energy are called fuels

Photo by Bruno in Unsplash (opens in a new tab)
Complete combustion of hydrocarbons and alcohols
When fuels like hydrocarbons and alcohols have a plentiful supply of oxygen, they undergo complete combustion
The carbon atoms in the fuel react with oxygen to form carbon dioxide (CO2)
The hydrogen atoms in the fuel react with oxygen to form water (H2O)
fuel + oxygen → carbon dioxide + water
Complete combustion word equations
Three examples of hydrocarbon and alcohol based fuels undergoing combustion are:
methane + oxygen → carbon dioxide + water
ethane + oxygen → carbon dioxide + water
ethanol + oxygen → carbon dioxide + water
Complete combustion chemical equations
You might be asked to balance chemical equations for combustion reactions
To balance combustion equations:
Balance the carbon atoms
Then, balance the hydrogen atoms
Finally, balance the oxygen atoms
Three examples of hydrocarbon and alcohol based fuels undergoing combustion are:
methane: CH4 + 2O2 → CO2 + 2H2O
ethane: 2C2H6 + 7O2 → 4CO2 + 6H2O
ethanol: C2H5OH + 3O2 → 2CO2 + 3H2O
Worked Example
Balance the symbol equation for the combustion of propane.
_C3H8 + _O2 → _CO2 + _H2O
[1]
Answer:
Balance the carbon atoms
There are 3 carbon atoms on the left hand side
So, place a 3 in front of carbon dioxide:
_C3H8 + _O2 → 3CO2 + _H2O
Then, balance the hydrogen atoms
There are 8 hydrogen atoms on the left hand side
So, place a 4 in front of water:
_C3H8 + _O2 → 3CO2 + 4H2O
Finally, balance the oxygen atoms
There are now 10 oxygen atoms on the right hand side
6 from carbon dioxide
4 from water
So, place a 5 in front of oxygen:
C3H8 + 5O2 → 3CO2 + 4H2O [1 mark]

Unlock more, it's free!
Was this revision note helpful?
