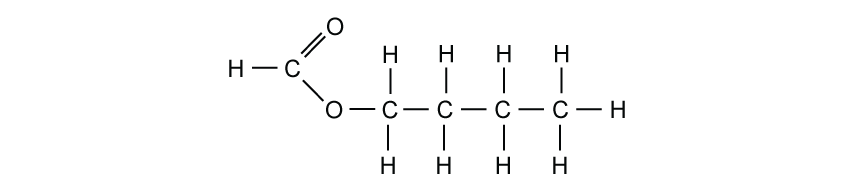
Which of these statements about carboxylic acids is not correct?
Aqueous ethanoic acid has a pH less than 7.
Methyl orange turns yellow in aqueous ethanoic acid.
They produce carbon dioxide when reacted with a metal carbonate.
They contain the functional group -COOH.
Was this exam question helpful?