Oxidation of Alcohols (AQA A Level Chemistry): Revision Note
Exam code: 7405
Oxidation of Alcohols
Oxidation of alcohols
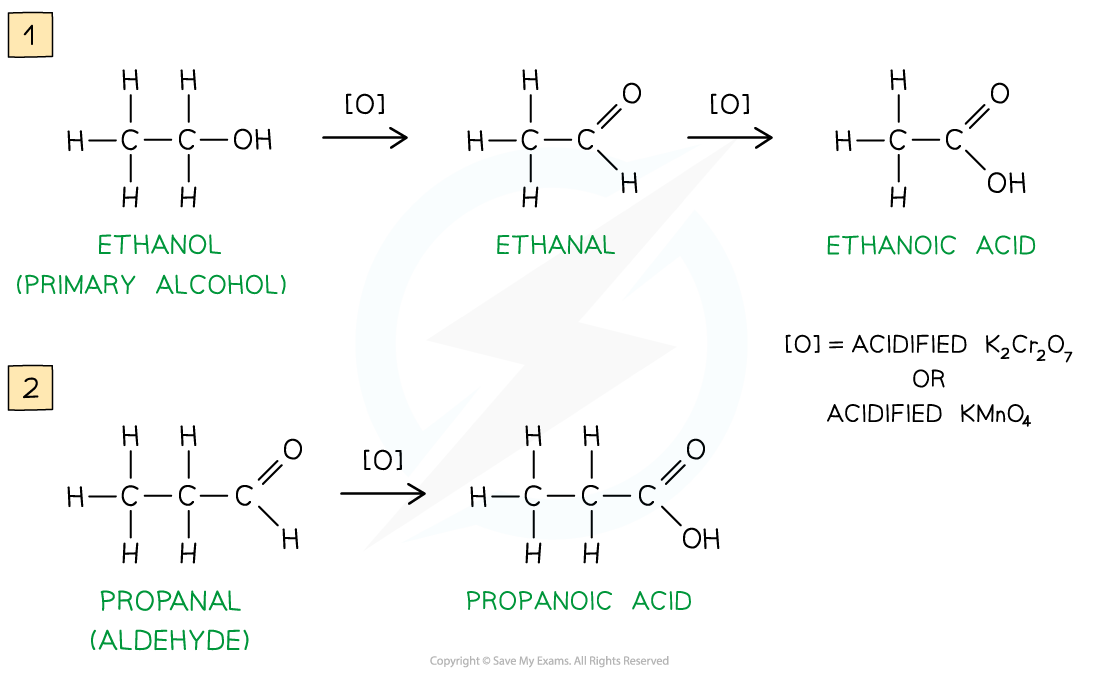
Primary alcohols can be oxidised to form aldehydes, which can undergo further oxidation to form carboxylic acids
Secondary alcohols can be oxidised to form ketones only
Tertiary alcohols do not undergo oxidation
Common oxidising agents for alcohols include acidified potassium dichromate(VI), K2Cr2O7 and acidified potassium manganate(VII), KMnO4
Oxidation Reaction Conditions
The primary alcohol is added to a limited amount of oxidising agent and gently warmed
The aldehyde product has a lower boiling point than the alcohol reactant, so it can be distilled off as soon as it forms.
If the aldehyde is not removed, further heating under reflux with excess oxidising agent will oxidise it to a carboxylic acid
Ketones, however, cannot be further oxidised, so the ketone product does not need to be distilled off immediately after it has formed
Simple Distillation

Heating Under Reflux
Heating under reflux is a laboratory technique used in chemistry to heat reactions at a constant temperature (boiling point) for extended periods without losing volatile solvent or reactants
A vertical condenser cools vapours, returning them to the flask
It is critical to never seal the top of the condenser to avoid pressure buildup and explosions.


Oxidation Products
Aldehydes and ketones are carbonyl compounds that contain a C=O group.
They can be prepared by oxidising primary and secondary alcohols, respectively.
Oxidising agents
Acidified potassium dichromate(VI)
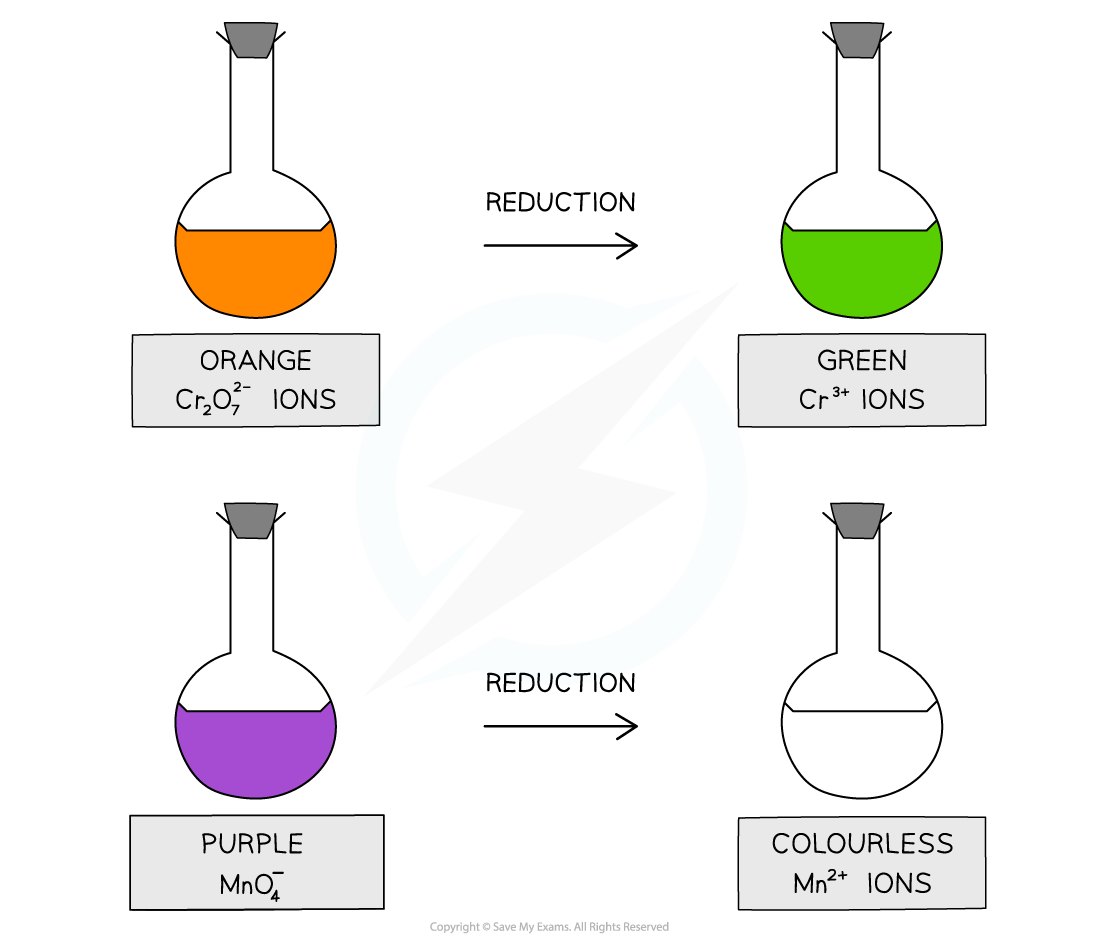
Acidified potassium dichromate(VI) is an orange oxidising agent
“Acidified” means that the dichromate is dissolved in a dilute acid, such as dilute sulfuric acid
For potassium dichromate(VI) to act as an oxidising agent, it must itself be reduced
This reduction requires hydrogen ions (H+), which are supplied by the acidic medium
When alcohols are oxidised, the orange dichromate ions (Cr2O72-) are reduced to green Cr3+ ions
Acidified potassium manganate(VII)
Acidified potassium manganate(VII) is a purple oxidising agent
As with potassium dichromate(VI), it must be in an acidic medium to allow reduction to occur
When alcohols are oxidised, the purple manganate ions (MnO4-) are reduced to colourless Mn2+ ions
Testing for Oxidation Products
The presence of an aldehyde group (-CHO) in an unknown compound can be determined by the oxidising agents Fehling’s and Tollens’ reagents
Fehling’s solution
Fehling’s solution is an alkaline solution containing copper(II) ions, which act as the oxidising agent
When warmed with an aldehyde, the aldehyde is oxidised to a carboxylic acid, and the Cu2+ ions are reduced to Cu+ ions
In the alkaline conditions, the carboxylic acid formed is neutralised to a carboxylate ion, as the –COOH group loses a proton to form –COO⁻
The carboxylate ion then forms a salt with a positively charged metal ion, such as sodium, producing a –COO-Na+ salt
During the reaction, the clear blue solution turns brick red due to the formation of a copper(I) oxide precipitate
Ketones cannot be oxidised and therefore give a negative result when warmed with Fehling’s solution

The colour changes in a Fehling's test
Tollens’ reagent
Tollens’ reagent is an aqueous alkaline solution of silver nitrate in excess ammonia and is also known as ammoniacal silver nitrate
When warmed in a water bath with an aldehyde, the aldehyde is oxidised to a carboxylic acid, and the Ag+ ions are reduced to silver atoms
In the alkaline conditions, the carboxylic acid formed is neutralised to a carboxylate ion and forms a salt
The silver atoms deposit on the inside of the test tube, forming a characteristic silver “mirror”
Ketones cannot be oxidised and therefore give a negative result when warmed with Tollens’ reagent


Unlock more, it's free!
Was this revision note helpful?
