Born-Haber Cycles (AQA A Level Chemistry): Revision Note
Exam code: 7405
Constructing Born-Haber Cycles
A Born–Haber cycle is a specific application of Hess’s Law used for ionic compounds. It allows the calculation of lattice enthalpy, which cannot be measured directly by experiment
The basic principle of constructing the cycle is to draw a diagram in which energy increases upwards

The cycle shows all the enthalpy changes required to convert elements into gaseous ions and then form the ionic lattice from those ions
The alternative route to the ionic lattice starts from the enthalpy of formation of the compound from its elements in their standard states
Drawing the cycle for sodium chloride
A good starting point is to draw the elements, with their state symbols, about one-third of the way up the diagram
These appear on the left-hand side of the equation for the process being represented
This position is marked by a horizontal line, which represents the starting energy level

Next, the gaseous ions must be formed
This occurs in two steps: first, gaseous atoms are produced, and then these atoms are converted into ions
The formation of gaseous atoms involves bond breaking, so these steps are represented by arrows drawn upwards on the diagram
It does not matter whether sodium or chlorine is considered first
The enthalpy of atomisation of sodium is:
Na (s) → Na (g) ΔHatꝋ = +108 kJ mol -1
The enthalpy of atomisation of chlorine is
½Cl2 (g) → Cl (g) ΔHatꝋ = +121 kJ mol -1
The products of each step are shown on horizontal lines, with the enthalpy change written next to the vertical arrow connecting the energy levels

Next, the gaseous ions are formed
The sodium atom loses an electron, so this energy change corresponds to the first ionisation energy of sodium:
Na (g) → Na+ (g) + e– ΔHieꝋ = +500 kJ mol-1
This process is endothermic, so the arrow is drawn upwards on the diagram
The chlorine atom gains an electron, which is the electron affinity of chlorine:
Cl (g) + e– → Cl- (g) ΔHeaꝋ = -364 kJ mol-1
This change is exothermic, so the arrow is drawn downwards
The electron affinity step is often displaced to the right on the diagram to improve clarity and avoid overlapping arrows

The two remaining parts of the cycle can now be completed.
The enthalpy of formation of sodium chloride is added at the bottom of the diagram:
Na(s) + ½Cl2 (g) → NaCl (s) ΔHfꝋ = -411 kJ mol -1
This is an exothermic change for sodium chloride, so the arrow is drawn downwards
Enthalpy of formation can be either exothermic or endothermic. If it is endothermic, the arrow must be drawn upwards and is often displaced to the right of the diagram for clarity
The final step is the lattice enthalpy, which can be shown as the enthalpy change for lattice formation or dissociation
For sodium chloride, the formation equation is:
Na+(g) + Cl-(g) → NaCl (s) ΔHlattꝋ

The cycle is now complete
It is most commonly used to calculate the lattice enthalpy of an ionic solid, but it can also be used to determine other enthalpy changes if the lattice enthalpy is known
Examiner Tips and Tricks
Lattice enthalpy can be defined as either the enthalpy of lattice dissociation or the enthalpy of lattice formation.
Lattice dissociation is a bond-breaking process, so the equation always shows the ionic solid changing into the gaseous ions in an endothermic reaction:
NaCl (s) → Na+(g) + Cl-(g) ΔHlattꝋ = + ve
Lattice formation is bond forming, so gaesous ions change into the ionic solid in an exothermic reaction
Na+(g) + Cl-(g) → NaCl (s) ΔHlattꝋ = - ve
Examiners use both processes in past paper questions.
Worked Example
Construct a Born-Haber Cycle which can be used to calculate the lattice energy of potassium chloride.
Step | Equation | Enthalpy Change |
|---|---|---|
Convert K(s) atoms into K(g) atoms | K (s) | |
Convert K(g) atoms into K+(g) ions | K (g) | |
Convert Cl2(g) molecules into Cl(g) atoms | ½Cl2 (g) | |
Convert Cl(g) atoms into Cl-(g) ions | Cl (g) + e- | |
Add up all values to get | ||
Apply Hess's Law to find |
|
Answer
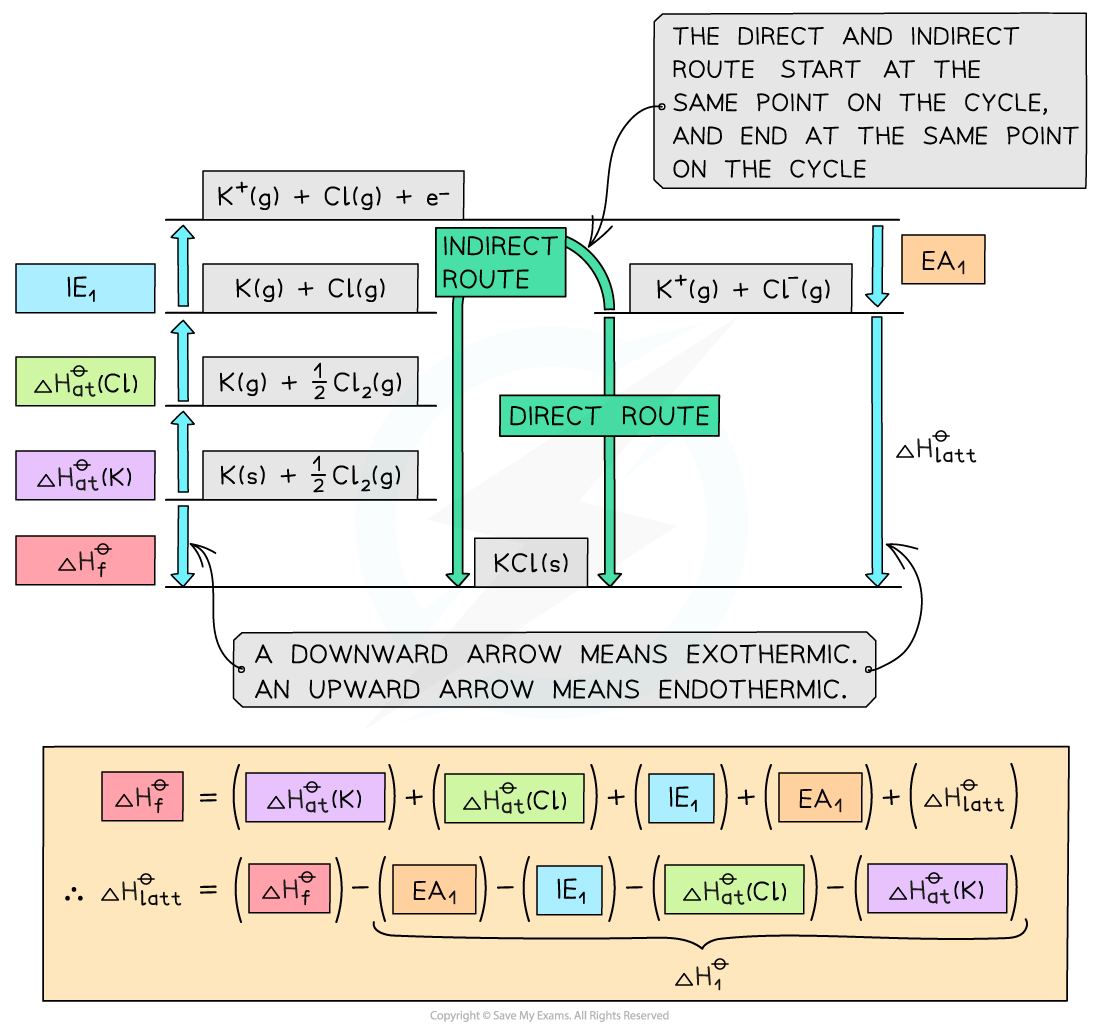
Worked Example
Construct a Born-Haber Cycle which can be used to calculate the lattice energy of magnesium oxide.
Convert Mg(s) atoms into Mg(g) atoms | Mg (s) | |
Convert Mg (g) atoms into Mg+(g) ions | Mg (g) | |
Convert Mg+(g) ions into Mg2+(g) ions | Mg+ (g) | |
Convert O2(g) molecules into O(g) atoms | ½O2 (g) | |
Convert O(g) atoms into O-(g) ions | O (g) + e- | |
Convert O-(g) ions into O2-(g) ions | O- (g) + e- | |
Add up all values to get | ||
Apply Hess's Law to find |
Answer
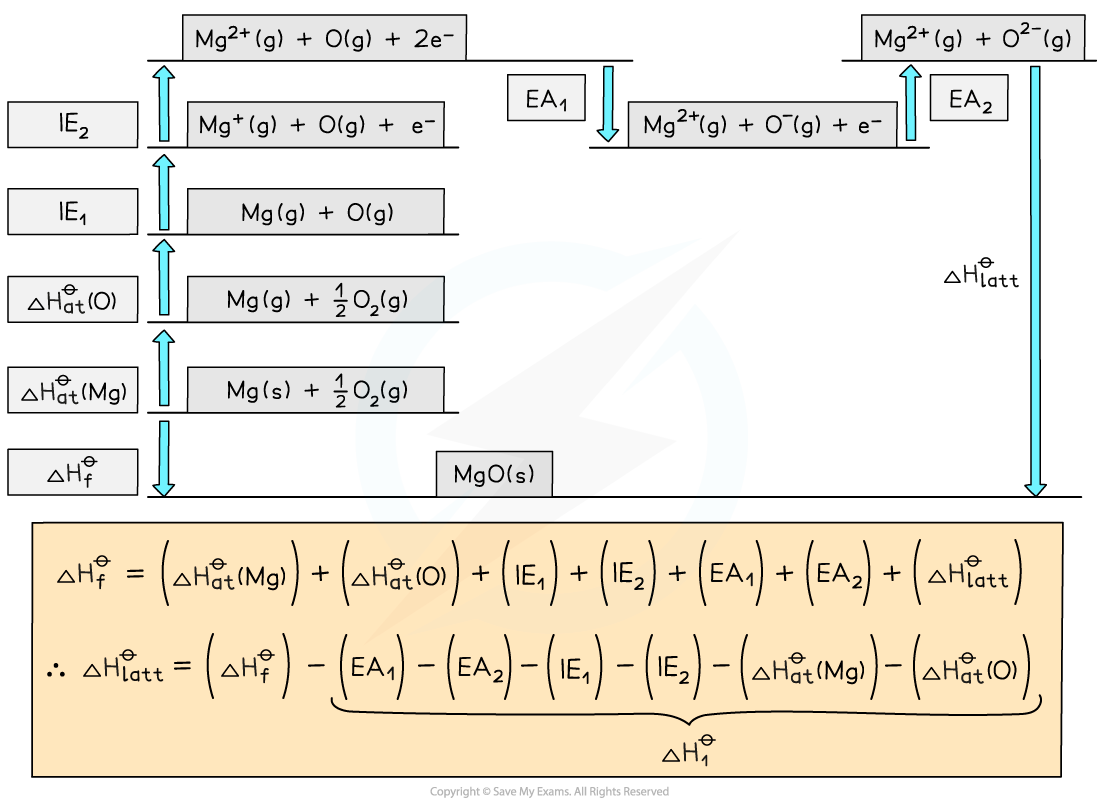
Examiner Tips and Tricks
When constructing Born–Haber cycles, the direction of each enthalpy change is important, but the relative size of the steps does not matter. Do not worry if the steps do not reflect the actual magnitudes of the energy changes.
You do not need to include an energy axis on a Born–Haber cycle. However, you must show the electron(s) in the ionisation step, as omitting them may result in the loss of marks.

Unlock more, it's free!
Was this revision note helpful?
