Finding Activation Energy (AQA A Level Chemistry): Revision Note
Exam code: 7405
Finding Activation Energy
The Arrhenius equation is commonly used to calculate the activation energy (Ea) of a reaction
The equation is:
k = A e−Ea/RT
Where
k is the rate constant,
A is the Arrhenius constant,
Ea is the activation energy,
R is the gas constant, and
T is the temperature in kelvin
In some cases, sufficient data are provided to substitute directly into the Arrhenius equation
Alternatively, a graph of ln k against 1/T can be plotted
The gradient of this straight-line graph is equal to −Ea/R, which can then be used to calculate the activation energy
Using the Arrhenius equation:
Remember, it is usually easier to use the version of the Arrhenius equation after natural logs of each side have been taken
Worked Example
Calculate the activation energy of a reaction which takes place at 400 K, where the rate constant of the reaction is 6.25 x 10-4 s-1.
A = 4.6 x 1013 and R = 8.31 J K-1 mol-1.
Answer:
Rearrange the equation for Ea:
Insert the values from the question:
Ea = 129095.85 J
Convert from J to kJ:
Ea = 129 kJ
Using an Arrhenius plot:
A graph of ln k against 1/T can be plotted, and then used to calculate Ea
This gives a line which follows the form y = mx + c
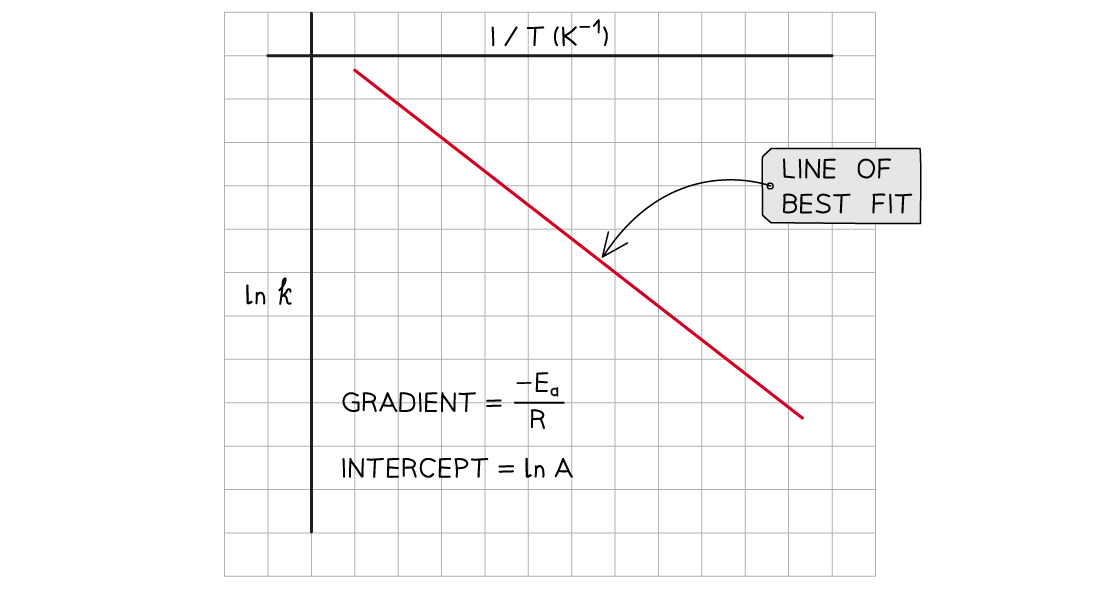
From the graph, the equation in the form of y = mx + c is:
Where:
y = ln k
x =
m =
(the gradient)
c = ln A (the y-intercept)
Worked Example
Complete the following table.
Temperature | 1/T | Time, t | Rate constant, k | ln k |
|---|---|---|---|---|
310 | 3.23 x 10-3 | 57 | -9.2 | |
335 | 31 | 3.01 x 10-4 | -8.1 | |
360 | 2.78 x 10-3 | 19 | 5.37 x 10-4 | -7.5 |
385 | 2.60 x 10-3 | 7 | 9.12 x 10-4 |
Plot a graph of ln k against 1/T.
Using the graph and the following equation, calculate the activation energy, Ea, for this reaction.
Calculate the Arrhenius constant, A, for this reaction.
Answers:
The completed table is:
Temperature | 1/T | Time, t | Rate constant, k | ln k |
|---|---|---|---|---|
310 | 3.23 x 10-3 | 57 | 1.01 x 10-4 | -9.2 |
335 | 2.99 x 10-3 | 31 | 3.01 x 10-4 | -8.1 |
360 | 2.78 x 10-3 | 19 | 5.37 x 10-4 | -7.5 |
385 | 2.60 x 10-3 | 7 | 9.12 x 10-4 | -7.0 |
The graph of ln k against 1/T is:
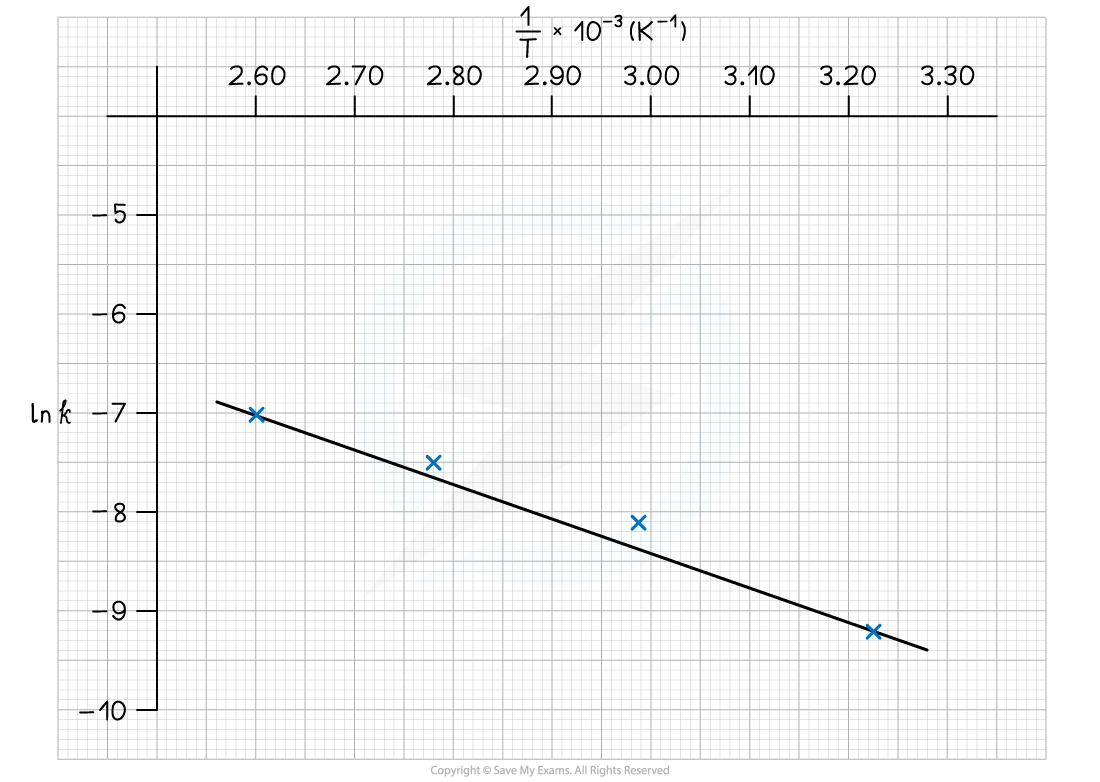
To calculate the activation energy, Ea:

Gradient = -Ea / R = -3666.6
Ea = -(-3666.6 x 8.31) = 30, 469 J mol-1
Ea = -(-3666.6 x 8.31) = 30.5 kJ mol-1
To calculate the Arrhenius constant, A:
Choose a point on the graph, e.g (2.60 x 10-3, -7.0)

Substitute values into the equation
-7.0 = (-3666.6 x 2.60 x 10-3) + ln A
-7.0 = -9.53 + ln A
ln A = 2.53
A =
A = 12.55
Examiner Tips and Tricks
You are not required to learn these equations. However, you do need to be able to rearrange them, and knowing them helps you understand the effects of temperature on the rate constant.

Unlock more, it's free!
Was this revision note helpful?
