Structure of Benzene (AQA A Level Chemistry): Revision Note
Exam code: 7405
Structure of Benzene
Aromatic Compounds
In everyday language, the word aromatic is used to describe pleasant or fragrant smells
In chemistry, however, aromatic refers to molecules that contain one or more benzene rings, meaning rings with conjugated π systems
Conjugated π systems arise from alternating single and double bonds, in which the π electrons are delocalised over the ring
Benzene is found in many useful compounds, including pharmaceuticals, pesticides, polymers, and dyes
Common painkillers such as aspirin, paracetamol, ibuprofen, and morphine all contain benzene rings
Examples of common aromatic compounds include:
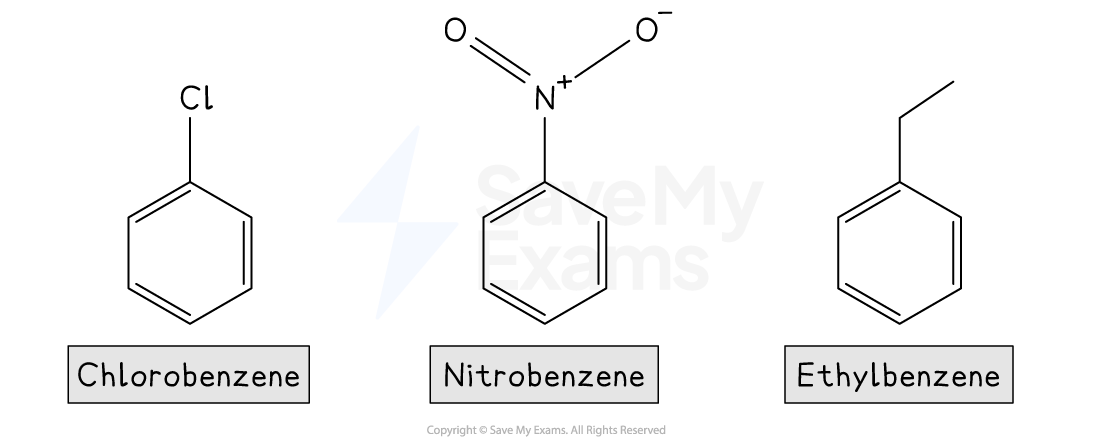
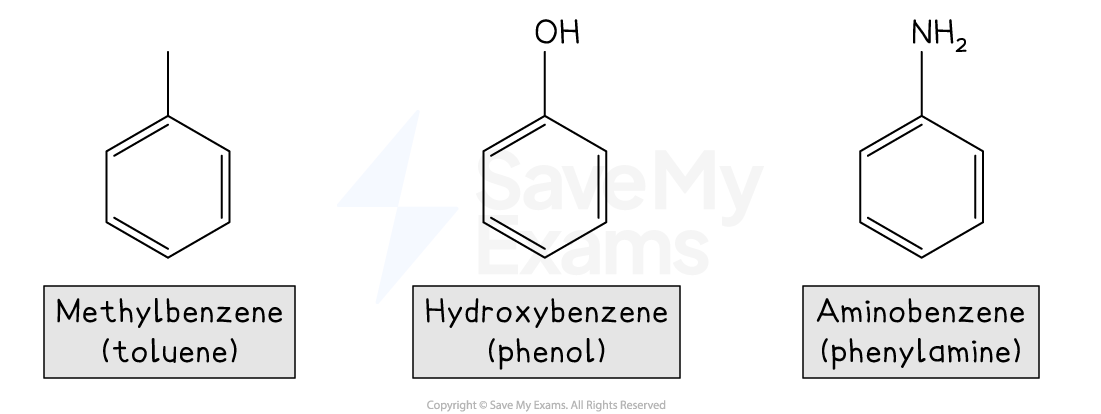
The Kekulé structure of benzene
The structure of benzene was proposed in 1865 by the Belgian chemist August Kekulé
Benzene consists of six carbon atoms arranged in a hexagonal ring, with alternating single and double carbon–carbon bonds.
This structure suggests that benzene should react like an unsaturated alkene

The Delocalisation Model
However, this is not the case, as Benzene and other aromatic compounds are regular and planar compounds with bond angles of 120 o
The delocalisation of electrons means that all of the carbon-carbon bonds in these compounds are identical and have both single and double bond character
The bonds all being the same length is evidence for the delocalised ring structure of benzene
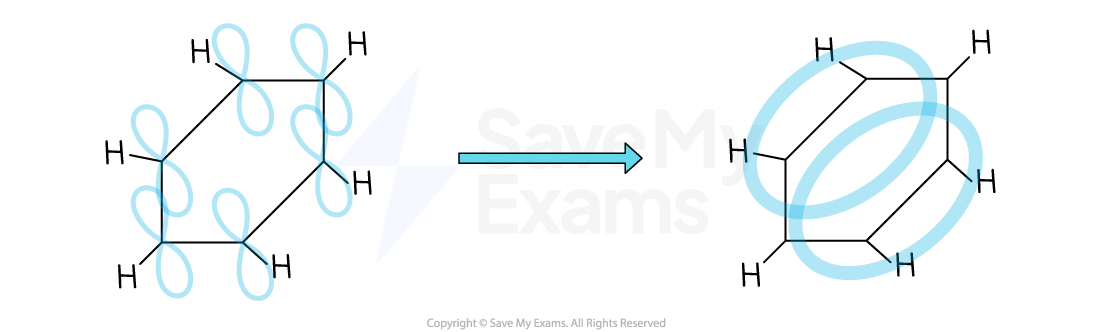
The delocalised model of benzene
Each carbon atom in the ring forms three σ bonds
The remaining p orbitals overlap laterally with the p orbitals of neighbouring carbon atoms to form a π system
This extensive sideways overlap of p orbitals results in the electrons being delocalised and able to spread over the entire ring
The π system is made up of two ring-shaped clouds of electron density - one above the plane and one below it

The modern representation of benzene is a hexagon with a circle inside
The circle represents the delocalised ring system
Electrophilic Substitution
Reactions of Benzene
The main reactions of benzene involve the replacement of one of the six hydrogen atoms on the benzene ring
This is different from the reactions of unsaturated alkenes, which involve breaking the C=C double bond and adding electrophile atoms across it
Reactions in which one or more hydrogen atoms in benzene are replaced are called electrophilic substitution reactions
In these reactions, a hydrogen atom is substituted by an electrophile
General Electrophilic Substitution Mechanism:


The delocalised π system in benzene is extremely stable and forms a region of high electron density
Electrophilic substitution reactions involve an electrophile, which may be a positively charged ion or the positive end of a polar molecule
Although many electrophiles are capable of reacting with benzene, they usually cannot be added directly to the reaction mixture
Instead, the electrophile must be generated in situ by adding appropriate reagents to the reaction mixture
Examiner Tips and Tricks
Make sure you understand the general steps of the electrophilic substitution mechanism and that you can explain what is happening - the same steps happen every time, the only difference is the electrophile used in the reaction!

Unlock more, it's free!
Was this revision note helpful?
