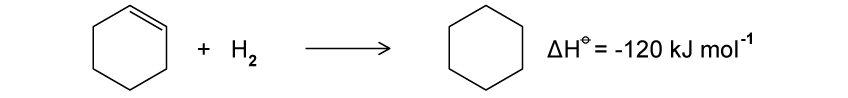
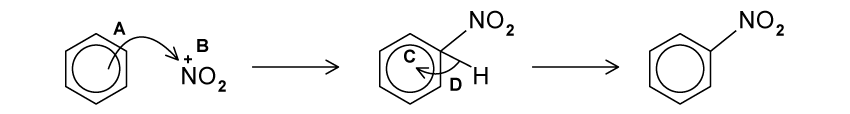
What reagents are required for the conversion of benzene to nitrobenzene?
Hydrogen gas in presence of nickel catalyst
Concentrated nitric acid and concentrated hydrochloric acid
Concentrated sulfuric acid and concentrated nitric acid
Tin with concentrated hydrochloric acid
Was this exam question helpful?