The rate of a reaction was monitored by recording the volume of gas produced every 5 seconds.

What is the mean rate of reaction in the first 10 seconds of the reaction, in cm3/s?
2.9
3.0
3.5
2.5
Was this exam question helpful?
Exam code: 0620 & 0971
Select a download format for Chemical Change & Rate of Reaction
Select an answer set to view for
Chemical Change & Rate of Reaction
Smart practice available on this topic
Answer questions with adaptive difficulty and personalised feedback
The rate of a reaction was monitored by recording the volume of gas produced every 5 seconds.

What is the mean rate of reaction in the first 10 seconds of the reaction, in cm3/s?
2.9
3.0
3.5
2.5
Choose your answer
Was this exam question helpful?
Which of the following processes represents a physical change?
Acid base neutralisation
Metal displacement reactions
Boiling ethanol
Combustion of magnesium
Choose your answer
Was this exam question helpful?
Physical and chemical changes are different processes. Which of the following rows correctly identifies characteristics of each one?
chemical change - no new substance formed
physical change - change of state
chemical change - no new substance formed
physical change - involves electron transfer
chemical change - new substance formed
physical change - involves electron transfer
chemical change - new substance formed
physical change - change of state
Choose your answer
Was this exam question helpful?
A student was measuring the rate of reaction using the apparatus in the diagram.

What reaction is the student studying?
CaO + H2SO4 → CaSO4 + H2O
Mg(OH)2 + 2HCl → MgCl2 + 2H2O
Fe + H2SO4 → FeSO4 + H2
KCl + AgNO3 → KNO3 + AgCl
Choose your answer
Was this exam question helpful?
A student was investigating the rate of reaction between iron and a solution of sulfuric acid. Two experiments were performed, P and Q, in which the volume of hydrogen was recorded at regular intervals.

Which change could explain the difference in results between P and Q?
The student added a catalyst in P.
Larger pieces of iron were used in P.
Less concentrated acid was used in P.
Larger quantities of both reactants are used P.
Choose your answer
Was this exam question helpful?
A student investigates the rate of reaction between aqueous sodium thiosulphate and aqueous hydrochloric acid by measuring the time taken for a precipitate to obscure a cross underneath the beaker.
Which statement is correct for the reaction?
The precipitate will be formed faster if ice is placed in contact with the beaker.
The precipitate will be formed faster if 10.0 cm3 of water are added to the aqueous sodium thiosulphate.
The precipitate will be formed slower if a catalyst is added to the reaction mixture.
The precipitate will be formed faster if drops of concentrated hydrochloric acid are added to the reaction mixture.
Choose your answer
Was this exam question helpful?
A student investigates the effect of temperature on the rate of a reaction.
In Experiment 1, the reaction is performed at 10 °C. In Experiment 2, the reaction is repeated at 20 °C. All other conditions stay the same.
Which graph shows the results for the volume of gas produced against time?
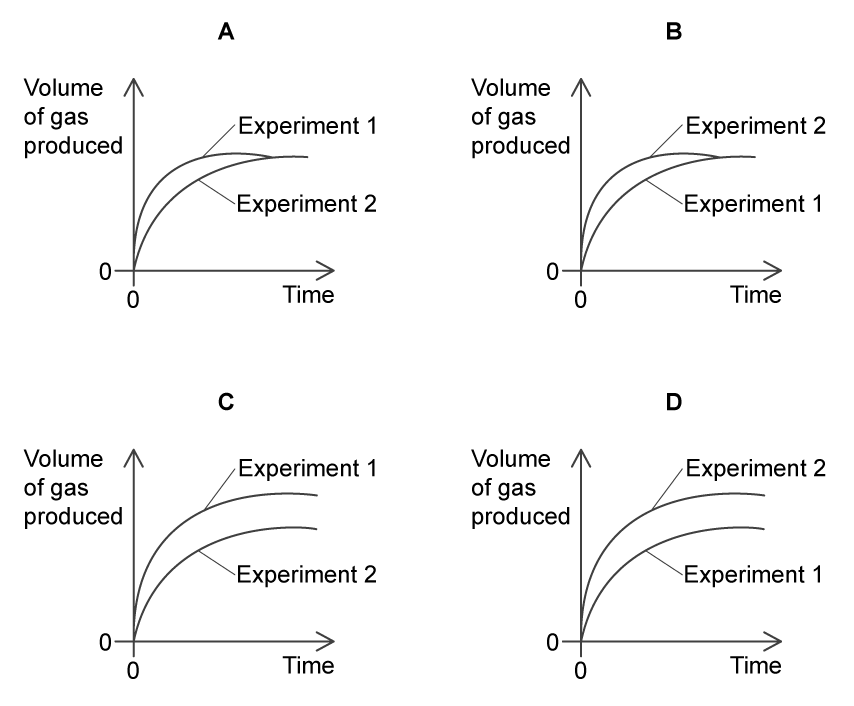
Choose your answer
Was this exam question helpful?
Which process is a physical change?
cracking of an alkane
hydration of ethene
fractional distillation of petroleum
thermal decomposition of calcium carbonate
Choose your answer
Was this exam question helpful?
A student was investigating the rate of reaction between a solid base and a solution of sulfuric acid. Two experiments were performed, S and T, in which the mass of the reaction flask was recorded as shown in the graph.

Which of the following changes could explain the difference in results between S and T?
The sulfuric acid is less concentrated in T.
The sulfuric acid is more concentrated in T.
A higher temperature is used in S.
Larger sized particles are used in T.
Choose your answer
Was this exam question helpful?
Which graph correctly shows the effect of increasing the temperature on the rate of reaction between magnesium and sulfuric acid in which neither reactant runs out?

Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
A student reacts a metal carbonate with nitric acid and measures the volume of carbon dioxide gas released at regular intervals. The results of the experiment are shown in the graph.

Between which two measurements was the reaction the fastest?
0 and 2 minutes
2 and 4 minutes
4 and 6 minutes
10 and 12 minutes
Choose your answer
Was this exam question helpful?
Magnesium metal reacts with an excess of hydrochloric acid solution to form magnesium chloride and hydrogen:
Mg (s) + 2HCl (aq) → MgCl2 (aq) + H2 (g)
Which of the following will not increase the rate of this reaction?
Increase the temperature of the acid
Increase the surface area to volume ratio of the pieces of magnesium
Increase the concentration of the hydrochloric acid
Increase the volume of hydrochloric acid solution used
Choose your answer
Was this exam question helpful?
Catalysts are often used in chemical reactions.
Which letter represents two correct features of catalysts?
Feature 1 - Lower activation energy for the reaction
Feature 2 - More products made in total
Feature 1 - Alternative pathway for the reaction is provided
Feature 2 - Not used up over the course of the reaction
Feature 1 - Makes industrial processes cheaper
Feature 2 - Metal catalysts are cheap to buy
Feature 1 - Reduce the overall energy change of the reaction
Feature 2 - Can be present in biological systems as enzymes
Choose your answer
Was this exam question helpful?
A student was investigating the rate of reaction between acid and metals. The experimental set-up is shown below.

Which test tube would give the quickest reaction?
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
The graph shows the results of two separate reaction rate investigations using excess calcium carbonate and sulfuric acid.

Which statement explains the difference between the results?
The temperature was higher in experiment 2.
Experiment 2 was allowed more time to react.
Same volume of a more concentrated acid was used in experiment 2.
More calcium carbonate was used in experiment 2.
Choose your answer
Was this exam question helpful?
Which of the following statements explains how increasing temperature increases the rate of a reaction?
Increasing the temperature increases the kinetic energy of the particles so there are a smaller number of successful collisions.
Increasing the temperature increases the kinetic energy of the particles so there are a larger number of unsuccessful collisions.
Increasing the temperature decreases the kinetic energy of the particles so there are a larger number of successful collisions.
Increasing the temperature increases the kinetic energy of the particles so there are a larger number of successful collisions.
Choose your answer
Was this exam question helpful?
Which of the following statements best describes the activation energy of a reaction?
The minimum energy released when colliding particles react.
The minimum energy required for particles to react when they collide.
The maximum energy required for particles to react when they collide.
The maximum energy released when colliding particles react.
Choose your answer
Was this exam question helpful?
Which of the following statements correctly describes the effects of increasing concentration on the rate of a reaction?
Increasing the concentration increases the kinetic energy of the particles, resulting in more successful collisions between particles per unit time.
Increasing the concentration increases the rate of reaction as there is more space between particles.
Increasing the concentration increases the rate of reaction as there are more collisions between particles per unit time.
Increasing the concentration increases the rate of reaction as there are more collisions between particles at the beginning of the reaction.
Choose your answer
Was this exam question helpful?
A student investigates the rate of reaction between sodium carbonate granules and dilute hydrochloric acid.
The investigation is carried out twice, as Experiment 1 and Experiment 2. The volume of acid, the temperature, and the mass of the sodium carbonate granules are kept the same in both experiments.
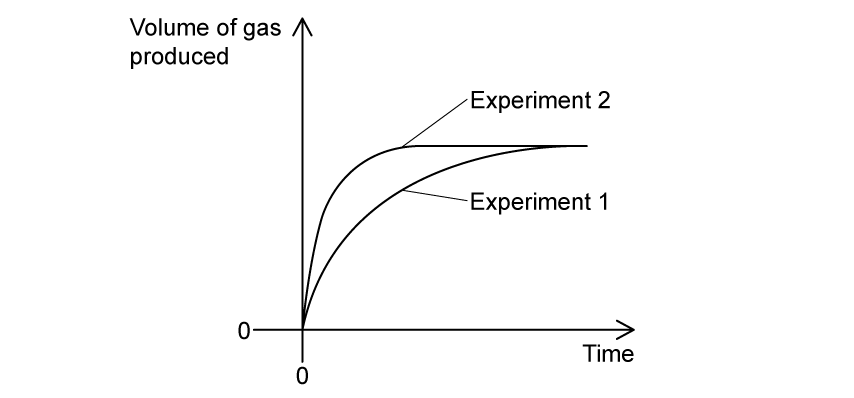
Which row correctly compares the concentration of the acid (C), the average kinetic energy of the particles (KE), and the frequency of successful collisions in the two experiments?
| average kinetic energy of particles | frequency of successful collisions | |
A | C2 > C1 | KE1 = KE2 | greater in experiment 2 |
B | C2 > C1 | KE2 > KE1 | greater in experiment 2 |
C | C1 > C2 | KE1 = KE2 | greater in experiment 1 |
D | C1 > C2 | KE2 > KE1 | greater in experiment 1 |
Choose your answer
Was this exam question helpful?