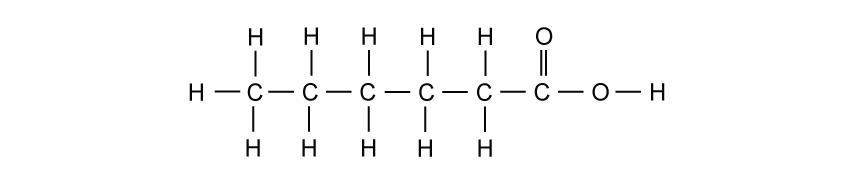
The alkanes are a family of saturated hydrocarbons. Their reactions include combustion, cracking and substitution.
i) What is meant by the term hydrocarbon?
[1]
ii) What is meant by the term saturated?
[1]
i) What is the general formula for the homologous series of alkanes?
[1]
ii) Calculate the mass of one mole of an alkane with 14 carbon atoms.
[2]
Cracking is used to obtain short-chain alkanes, alkenes and hydrogen from long-chain alkanes.
i) Give a use for each of the three products listed above.
short-chain alkanes ..................................................
alkenes ..................................................
hydrogen ..................................................
[3]
ii) Write an equation for the cracking of decane, C10H22, which produces two different alkenes and hydrogen as the only products.
[1]
Separate: Chemistry and Extended Only
Chlorine reacts with propane in a substitution reaction to form 1-chloropropane.
CH3–CH2–CH3 + Cl2 → CH3–CH2–CH2–Cl + HCl
i) What is the essential condition for the above reaction?
[1]
ii) There is more than one possible substitution reaction between chlorine and propane.
Suggest the structural formula of a different product.
[1]
Was this exam question helpful?