Which metal could not be used for electroplating by using an aqueous solution?
Chromium
Copper
Silver
Sodium
Was this exam question helpful?
Exam code: 0620 & 0971
Select a download format for Applications of Electrolysis
Select an answer set to view for
Applications of Electrolysis
Smart practice available on this topic
Answer questions with adaptive difficulty and personalised feedback
Which metal could not be used for electroplating by using an aqueous solution?
Chromium
Copper
Silver
Sodium
Choose your answer
Was this exam question helpful?
During electroplating what are the correct anode (positive electrode) and cathode (negative electrode) products when aqueous copper(II) sulfate is electrolysed using copper electrodes?
| Anode product | Cathode product |
A | copper(II) ions | copper metal |
B | copper(II) ions | hydrogen gas |
C | oxygen gas | copper metal |
D | oxygen gas | hydrogen gas |
Choose your answer
Was this exam question helpful?
In a fuel cell ............1........... energy is converted to ..................2................ energy.
| 1 | 2 |
A | electrical | heat |
B | chemical | electrical |
C | potential | chemical |
D | chemical | heat |
Choose your answer
Was this exam question helpful?
Hydrogen-oxygen fuel cells can be used to power motor vehicles.
What is the correct balanced symbol equation for the reaction that occurs in a hydrogen-oxygen fuel cell?
H2O → H2 + O2
H2 + O2 → 2H2O
2H2 + O2 → 2H2O
H2 + O2 → H2O
Choose your answer
Was this exam question helpful?
A student sets up the following equipment to investigate electroplating.

What would they see occur during the electroplating process?
The anode would be covered with an orange-pink coating
The cathode would be covered with a silver coating
The cathode would be covered with an orange-pink coating
The anode would be covered with a silver coating
Choose your answer
Was this exam question helpful?
An iron tray is electroplated with pure silver to improve its appearance. The diagram shows the electrochemical cell with labels X, Y and Z.
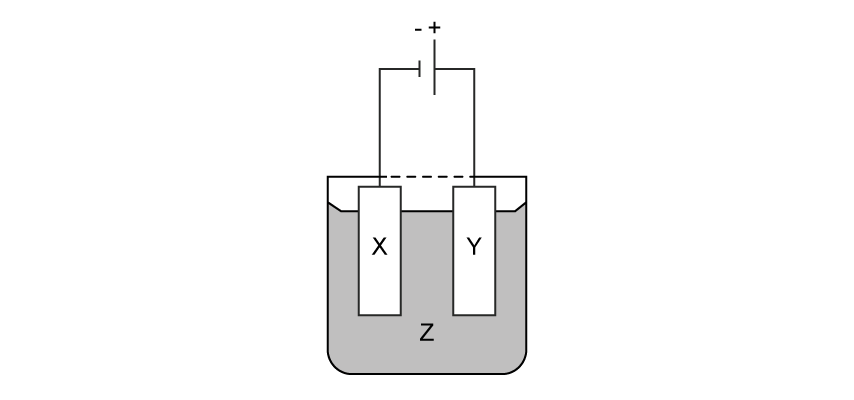
Which row correctly identifies X, Y, and the electrolyte Z?
Electrode X (at the negative terminal) | Electrode Y (at the positive terminal) | Electrolyte Z | |
A | Pure silver | Iron tray | Silver nitrate |
B | Pure silver | Iron tray | Iron (II) chloride |
C | Iron tray | Pure silver | Silver nitrate |
D | Iron tray | Pure silver | Silver chloride |
Choose your answer
Was this exam question helpful?
Three statements comparing gasoline (petrol) combustion engines with hydrogen-oxygen fuel cells are shown
The combustion of gasoline and the formation of water are both exothermic processes with a positive enthalpy change, ΔH.
Combustion engines are more polluting than fuel cells because incomplete combustion produces carbon monoxide and particulates.
Gasoline is more difficult and expensive to store in a vehicle than hydrogen.
Which statements are correct?
1 and 2
2 only
2 and 3
1, 2 and 3
Choose your answer
Was this exam question helpful?
The diagram shows a failed attempt to copper-plate a pan.

Which can be done to plate the pan with copper?
Cool the copper sulfate solution in an ice bath.
Heat the copper sulfate solution to boiling point.
Increase the voltage from 3 V to 6 V.
Make the pan the cathode and the copper the anode.
Choose your answer
Was this exam question helpful?
Which of the following are advantages of a hydrogen fuel cell to power a car?
No power is lost on transmission as there are less moving parts than in an internal combustion engine.
Power is lost on transmission as there are more moving parts than in an internal combustion engine.
They produce carbon dioxide.
They produce oxides of nitrogen.
Choose your answer
Was this exam question helpful?
The diagram shows a simple cell.

Which statement about the process occurring when the cell is in operation is correct?
Cu2+ ions are formed in solution.
Electrons travel through the solution.
The reaction Zn → Zn2+ + 2e– occurs.
The zinc electrode increases in mass
Choose your answer
Was this exam question helpful?
A students wants to electroplate an iron spoon with silver using an aqueous silver salt as the electrolyte.
Which set of conditions is used?
| Iron spoon | Other electrode |
A | cathode | silver |
B | anode | silver |
C | cathode | carbon |
D | anode | carbon |
Choose your answer
Was this exam question helpful?
What are the products made by a hydrogen-oxygen fuel cell?
1 Water
2 Oxygen
3 Hydrogen
1 only
1 and 2
2 and 3
1, 2 and 3
Choose your answer
Was this exam question helpful?
A student wishes to electroplate an object with copper.
Which row is correct?
| object is made at the | a suitable electrolyte is |
A | anode | CuO(s) |
B | anode | CuSO4(aq) |
C | cathode | CuO(s) |
D | cathode | CuSO4(aq) |
Choose your answer
Was this exam question helpful?
Winston Churchill, a British Prime Minister, had his false teeth electroplated with gold.
The teeth were coated with a thin layer of carbon and were then placed in the apparatus shown.

| terminal X is | the carbon powder could be |
A | negative | diamond |
B | negative | graphite |
C | positive | diamond |
D | positive | graphite |
Choose your answer
Was this exam question helpful?
The diagram shows an electrolysis experiment using X and Y as electrodes.

Copper is used to coat the other metal.
Which metal becomes coated and which solution is used?
| aqueous solution | metal |
A | CoCl3 | Y |
B | CuCl2 | Y |
C | CuCl2 | X |
D | CoCl3 | X |
Choose your answer
Was this exam question helpful?
What is the correct balanced symbol equation for the overall reaction in a hydrogen-oxygen fuel cell?
2H2 → 4H+ + 4e–
4H+ + O2 + 4e– → 2H2O
2H2 + O2 → 2H2O
2H2O → 2H2 + O2
Choose your answer
Was this exam question helpful?
The following apparatus for plating a spoon with silver is shown.

Which statements are correct?
1 The metal electrode would be connected to the negative terminal of the power supply
2 The spoon would be connected to the negative terminal of the power supply
3 At the cathode, silver ions lose electrons to form silver atoms
4 At the anode, silver atoms lose electrons to form silver ions
2 only
1 and 4 only
2 and 4 only
1, 2 and 3
Choose your answer
Was this exam question helpful?
An iron tray is electroplated with pure silver to improve its appearance. The diagram shows the electrochemical cell with labels X, Y and Z.

Which row correctly identifies the half-equations occurring at electrodes X and Y, and the identity of electrolyte Z?
half-equation at X | half-equation at Y | electrolyte Z | |
A | Ag+ + e− → Ag | Ag → Ag++ e− | aqueous silver nitrate |
B | Ag → Ag++ e− | Ag+ + e− → Ag | aqueous silver nitrate |
C | Ag+ + e− → Ag | Fe2+ + 2e− → Fe | aqueous iron(II) nitrate |
D | Fe2+ + 2e− → Fe | Ag → Ag++ e− | aqueous silver nitrate |
Choose your answer
Was this exam question helpful?