Which of the following formulae shows an alkane?
C2H4
C4H8
C4H10
C5H10
Was this exam question helpful?
Exam code: 0620 & 0971
Select a download format for Formulae, Functional Groups & Terminology
Select an answer set to view for
Formulae, Functional Groups & Terminology
Smart practice available on this topic
Answer questions with adaptive difficulty and personalised feedback
Which of the following formulae shows an alkane?
C2H4
C4H8
C4H10
C5H10
Choose your answer
Was this exam question helpful?
Which of the following structures shows a compound that belongs to a different homologous series to octane?

Choose your answer
Was this exam question helpful?
The diagram shows the structure of ethene.

Which of these compounds has similar chemical properties of ethene?

Choose your answer
Was this exam question helpful?
The structures of four organic compounds are shown.
Which one does not show the displayed formula of the compound?

Choose your answer
Was this exam question helpful?
Which of the following is not a feature of a homologous series?
Compounds have the same functional group
Compounds have the same physical properties
Compounds have the same general formula
Compounds have similar chemical properties
Choose your answer
Was this exam question helpful?
Which diagram shows the correct displayed formula for butanoic acid?
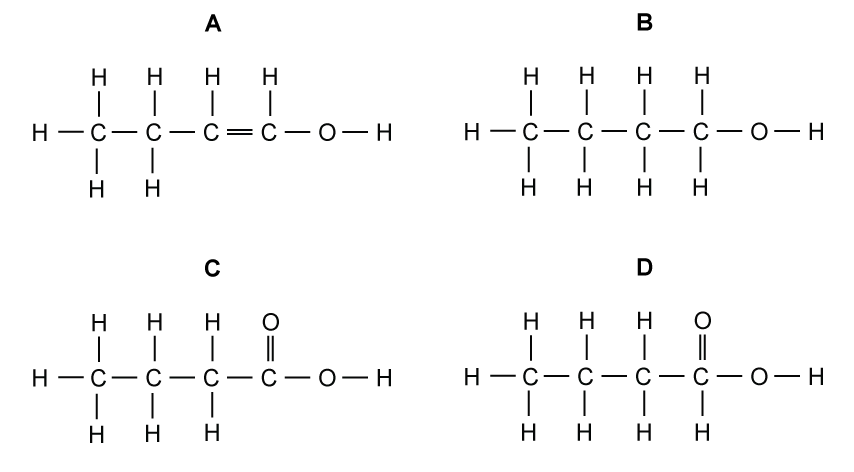
Choose your answer
Was this exam question helpful?
Which one of the following structures is correctly named?

Choose your answer
Was this exam question helpful?
Organic compounds can have names that have the suffixes of -ane, -ene, -ol or -oic acid.
How many of these have at least one double bond in their structure
1
2
3
4
Choose your answer
Was this exam question helpful?
When the number of atoms in a molecule of a hydrocarbon increases, the amount of energy released when it burns increases.
What is the correct order?
| less energy | → | more energy released |
A | methane | ethene | ethane |
B | ethene | ethane | methane |
C | ethene | methane | ethane |
D | methane | ethane | ethene |
Choose your answer
Was this exam question helpful?
The diagram below represents the molecule of a compound.

What is the name of this compound?
Ethane
Ethanol
Ethanoic acid
Ethene
Choose your answer
Was this exam question helpful?
The structures of six organic compounds compound A are shown.

Which two compounds are isomers of one another?
B and D
A and C
C and F
C and E
Choose your answer
Was this exam question helpful?
Three statements about propane and propanol are listed below.
Propane and propanol belong to the same homologous series
Propanol is a compound that contains three carbon atoms
Propane is an unsaturated hydrocarbon
Which statements are correct?
1 and 2 only
1 and 3 only
2 only
3 only
Choose your answer
Was this exam question helpful?
The first three members of a homologous series are shown.

Why do these molecules represent a homologous series?
They contain carbon and hydrogen atoms.
They have saturated bonds.
They have the same functional group.
They react differently from each other.
Choose your answer
Was this exam question helpful?
Which pair of the following structures shows a homologous series?

Choose your answer
Was this exam question helpful?
Which group of compounds is from a homologous series?
CH3OH, C2H5OH, C3H7OH
C3H6, C3H8, C3H7OH
CH4, C2H4, C3H8
CH3CO2H, CH3CH2OH, HCO2H
Choose your answer
Was this exam question helpful?
The diagram below shows some simple hydrocarbons and the products of two reactions.

Which of these structures are named correctly?
| Structure 1 | Structure 2 | Structure 3 |
A | x | ✔ | ✔ |
B | ✔ | ✔ | ✔ |
C | x | ✔ | x |
D | ✔ | x | ✔ |
Choose your answer
Was this exam question helpful?
The diagram below shows a reaction scheme. What are the names of all of the compounds?

| P | Q | R | S |
A | ethene | ethane | ethanoic acid | ethanol |
B | ethane | ethene | ethanol | ethanoic acid |
C | ethene | ethane | ethanol | ethanoic acid |
D | ethane | ethene | ethanoic acid | ethanol |
Choose your answer
Was this exam question helpful?
The displayed formula of an organic compound is shown.
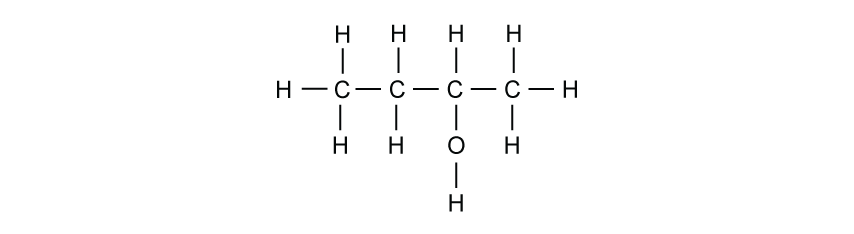
What is the name of this compound?
butan-1-ol
butan-2-ol
butan-3-ol
butanol
Choose your answer
Was this exam question helpful?
W, X, Y and Z are the first four members of the carboxylic acid homologous series, but not necessarily in that order.
The table shows their relative molecular masses, Mr.
substance | relative molecular mass, Mr |
W | 60 |
X | 88 |
Y | 46 |
Z | 74 |
Which sequence shows the substances in order of decreasing boiling point?
X → Z → W → Y
Y → W → Z → X
W → X → Y → Z
Z → X → W →Y
Choose your answer
Was this exam question helpful?