Which diagram shows an alloy?

Was this exam question helpful?
Exam code: 0620 & 0971
Select a download format for Properties, Uses & Alloys of Metals
Select an answer set to view for
Properties, Uses & Alloys of Metals
Smart practice available on this topic
Answer questions with adaptive difficulty and personalised feedback
Which diagram shows an alloy?

Choose your answer
Was this exam question helpful?
Copper is sometimes used to make cooking utensils and pans.
Three properties of copper are shown below:
1 | Good conductor of electricity. |
2 | Corrosion resistant. |
3 | Good conductor of heat. |
Which of these properties make it good for making cooking utensils?
1 and 3
1 only
2 and 3
2 only
Choose your answer
Was this exam question helpful?
Most of the elements on the Periodic Table are metals. Metals can be distinguished from non-metals by differences in their physical properties.
Which row correctly describes a metal?
| Malleability | Heat & electrical conductivity |
A | ||
B | ||
C | ||
D |
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
Zinc has many uses in the home. The diagram shows two items that contain zinc.

In which of these items is zinc used as an alloy?
| bucket | door-knocker |
A | ||
B | ||
C | ||
D |
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
Why are alloys harder than the pure metals from which they are made?
There are stronger bonds between the atoms
The atoms are all the same size
The layers of atoms are distorted
The properties of each element have combined
Choose your answer
Was this exam question helpful?
Steel is an alloy of iron and carbon.
Which diagram represents steel correctly?
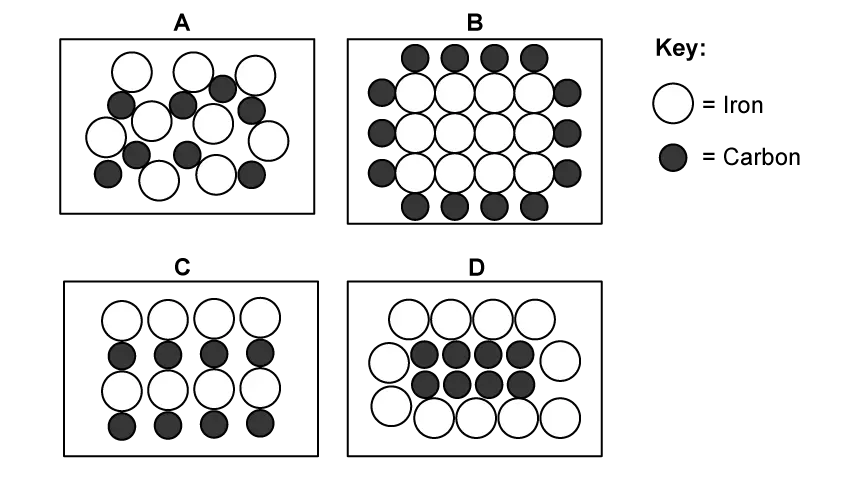
Choose your answer
Was this exam question helpful?
Three statements about metals are listed.
Aluminium is used for aircraft because of its high resistance and high density.
Metals react with acids to form salt and water.
Copper is used in electrical wires because of its malleable and a good electricity conductor
Which statements are correct?
1, 2 and 3
1 and 3 only
3 only
1 and 2 only
Choose your answer
Was this exam question helpful?
A sample of element Y is shiny and can be shaped by hammering.
Which row of the table corresponds to the properties of element Y?
| conducts electricity | melts below 25℃ |
A | ||
B | ||
C | ||
D |
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
Some of the properties of four different elements Q, P, R and S are listed below:
1 | Q melts at 1410 °C and forms an acidic oxide. |
2 | P has a high density and is easily drawn into wires. |
3 | R acts as a catalyst and its oxide reacts with acids |
4 | S is a red-brown solid used to make alloys. |
Which of the elements are metals?
1 and 3 only
2 and 4 only
2, 3 and 4
1,2 and 3
Choose your answer
Was this exam question helpful?
The table shows some properties of four metals
Which of the four metals would be the most suitable for aircraft construction?
| density | strength | resistance to corrosion |
A | high | low | low |
B | low | high | high |
C | high | high | low |
D | low | low | high |
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
Brass is an alloy of two metals.
Which row describes the correct use of the two metals that brass is made from?
| metal 1 | metal 2 |
A | galvanising steel | making aircraft |
B | electrical wiring | galvanising steel |
C | making aircraft | making cutlery |
D | making cooking pans | electrical wiring |
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
The structure of an alloy is shown below. Alloys are mixtures of metals with other metals or non-metals.

They are usually much stronger and harder than pure metals. Which statement explains these features?
The irregular sizes of the atoms distorts the regular arrangement in the metal structure, meaning more force is required to bend the metal
The irregular sizes of the atoms distorts the regular arrangement in the metal structure as the bonding is disrupted when the atoms are added
The regular sizes of the atoms distorts the irregular arrangement in the metal structure, meaning more force is required to bend the metal
A mixture of metals is tougher than a pure metal because a mixture has more intermolecular forces than a pure metal.
Choose your answer
Was this exam question helpful?
Iron is a transition element and magnesium is a Group II element. Both are metals.
Which row shows the correct properties for both metals?
melting point | gas produced when reacted with dilute hydrochloric acid | electrical conductivity | |
A | iron > magnesium | hydrogen | good |
B | iron > magnesium | chlorine | good |
C | iron = magnesium | hydrogen | poor |
D | iron = magnesium | chlorine | poor |
Choose your answer
Was this exam question helpful?
Which statement explains why alloys are harder and stronger than pure metals?
Alloys contain strong covalent bonds between the different atoms.
The metallic bonds in an alloy are stronger because there are more delocalised electrons.
The atoms in an alloy are packed more closely together than in a pure metal.
The different sized atoms in an alloy mean the layers can no longer slide over each other.
Choose your answer
Was this exam question helpful?
Properties of four elements are shown below.
Which of these elements is a metal?
| electrical conductivity when solid | electrical conductivity when liquid | melting point ℃ |
A | low | low | -7 |
B | high | low | 801 |
C | high | high | 1535 |
D | low | low | 3550 |
Option A
Option B
Option C
Option D
Choose your answer
Was this exam question helpful?
Brass is an alloy of two metals
Which is the correct statement?
The alloy will completely dissolve in dilute hydrochloric acid.
Brass is formed by a chemical reaction between two metals
Brass can be represented by a chemical formula
One of the metals in the alloy will dissolve in dilute hydrochloric acid
Choose your answer
Was this exam question helpful?
Element Y is malleable and ductile.
Which correctly describes the properties of element Y?
| Electrical conductivity (solid) | Electrical conductivity (molten) | Melting Point / oC |
1 | ✓ | ✓ | 2861 |
2 | ✓ | ✓ | 63 |
3 | ✗ | ✓ | 801 |
1 and 2 only
1 and 3 only
2 and 3 only
1, 2 and 3
Choose your answer
Was this exam question helpful?
The three statements below about aluminium are all correct.
Aluminium reacts readily to form aluminium oxide.
Aluminium is extracted from its ore by electrolysis as it cannot be extracted by reduction with carbon.
Aluminium is used in electrical wiring because of its good electrical conductivity.
In which of these statements can 'aluminium' be replaced by 'copper' and still be correct?
| 1 | 2 | 3 |
A | ✓ | ✓ | ✓ |
B | ✗ | ✓ | ✗ |
C | ✓ | ✗ | ✓ |
D | ✗ | ✗ | ✓ |
Choose your answer
Was this exam question helpful?
Some properties of elements K, L, M and N are given.
1 | K acts as a catalyst and its oxide reacts with hydrochloric acid |
2 | L is a dull dark grey solid that conducts electricity and has a melting point of 3600 oC |
3 | M is easily hammered into shape and has a high density |
4 | N has a melting point of 113 oC and forms an acidic oxide |
Which of the elements are metals?
1 and 3 only
2, 3 and 4
2 and 3 only
2 and 4 only
Choose your answer
Was this exam question helpful?