Which option correctly describes the relative charges and masses of the subatomic particles?
| Proton | Neutron | Electron | Relative mass of proton |
A | +1 | 0 | 0 | 0.00054 |
B | 0 | +1 | +1 | 1 |
C | 0 | +1 | -1 | 0.00054 |
D | +1 | 0 | -1 | 1 |
Was this exam question helpful?
Exam code: 0620 & 0971
Select a download format for Atomic Structure & the Periodic Table
Select an answer set to view for
Atomic Structure & the Periodic Table
Smart practice available on this topic
Answer questions with adaptive difficulty and personalised feedback
Which option correctly describes the relative charges and masses of the subatomic particles?
| Proton | Neutron | Electron | Relative mass of proton |
A | +1 | 0 | 0 | 0.00054 |
B | 0 | +1 | +1 | 1 |
C | 0 | +1 | -1 | 0.00054 |
D | +1 | 0 | -1 | 1 |
Choose your answer
Was this exam question helpful?
The atomic number of element Q is 9 and its mass number is 19.
In which group of the Periodic Table should element Q be placed?
Group 0
Group I
Group VII
Group II
Choose your answer
Was this exam question helpful?
The diagram shows the atomic structure of an element.

Which element is it?
Carbon
Beryllium
Oxygen
Boron
Choose your answer
Was this exam question helpful?
The atomic structure of atoms Q, R, S and T are shown in the table.
Atom | Number of protons | Number of neutrons | Number of electrons |
Q | 6 | 7 | 6 |
R | 7 | 7 | 7 |
S | 6 | 8 | 6 |
T | 8 | 8 | 8 |
Which two atoms are isotopes?
Q and S
Q and T
R and T
R and S
Choose your answer
Was this exam question helpful?
Substances can be broadly classified into three categories which are elements, compounds and mixtures.
Which of the following statements is correct?
Elements contain different atoms
Compounds consist of elements that are not chemically combined
Mixtures consist of elements that are chemically combined
A mixture can contain elements and compounds
Choose your answer
Was this exam question helpful?
Which diagram shows a mixture of compounds?
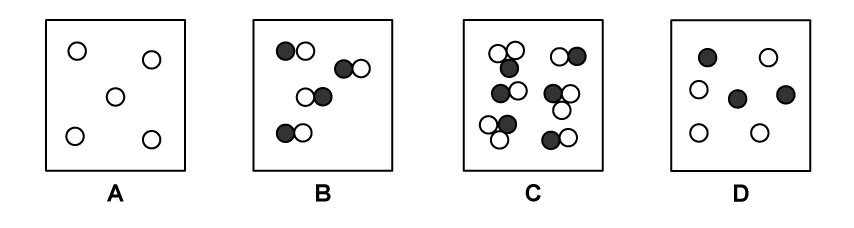
Choose your answer
Was this exam question helpful?
Which statement about carbon is correct?
It has 4 electron shells.
It has 2 outer shell electrons.
It has 12 nucleons.
It has an electron configuration of 2,8,2.
Choose your answer
Was this exam question helpful?
Two atoms, X and Y can be represented as shown
Which row is correct for both atoms?
electron configuration of X | electron configuration of Y | Is Y an isotope of X? | |
A | 2,1 | 2,1 | Yes |
B | 2,1 | 2,6 | Yes |
C | 2,5 | 2,1 | No |
D | 2,5 | 2,6 | No |
Choose your answer
Was this exam question helpful?
The structure of four particles is described in the table.
Particle | Number of protons | Number of neutrons | Number of electrons |
Li | 3 | X | 3 |
Li+ | 3 | 4 | 2 |
F | Y | 10 | 9 |
F- | 9 | 10 | Z |
What are the correct values for X, Y and Z?
| X | Y | Z |
A | 4 | 7 | 9 |
B | 3 | 9 | 9 |
C | 4 | 9 | 10 |
D | 3 | 7 | 10 |
Choose your answer
Was this exam question helpful?
Element X contains 12 of each sub-atomic particle.
What is the correct Group number for element X?
Group 0
Group I
Group VII
Group II
Choose your answer
Was this exam question helpful?
Element X contains 4 electrons in its outer shell and 125 neutrons in its nucleus. What is element X?
Beryllium
Carbon
Tin
Lead
Choose your answer
Was this exam question helpful?
Potassium fluoride is an ionic compound formed when one atom of potassium bonds with one atom of fluorine.
Which row correctly describes the charge and electronic configuration of each ion in the compound?
| Potassium ion | Fluorine ion | ||
A | 1 - | 2, 8, 8 | 1 + | 2, 8 |
B | 1 + | 2, 8, 8 | 1 - | 2, 8 |
C | 1 + | 2, 8, 8 ,1 | 1 - | 2, 7 |
D | 1 - | 2, 8, 8, 2 | 1 + | 2, 6 |
Choose your answer
Was this exam question helpful?
A section of the Periodic Table is shown below.

The electronic structure of elements W, X, Y and Z are shown in the table.
| Electronic structure |
W | 2, 1 |
X | 2, 4 |
Y | 2, 8, 2 |
Z | 2, 8 |
Which elements have the correct electronic structure?
W and X
Y and Z
W and Y
X and Z
Choose your answer
Was this exam question helpful?
Which diagram shows a mixture of noble gases?
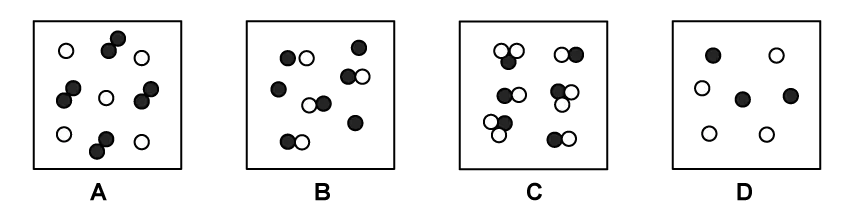
Choose your answer
Was this exam question helpful?
Two atoms, X and Y can be represented as shown
Which statement is correct for both atoms?
They have the same chemical properties because they have the same number of neutrons.
They have the same chemical properties because they have the same number of electrons in their outer shell.
They have the same physical properties because they have the same number of electrons in their outer shell.
They have the same physical properties because they have the same number of protons.
Choose your answer
Was this exam question helpful?
The X+ ion of element X has 16 electrons and a nucleon number of 37.
What is the correct Group number for element X?
VII
VIII
III
I
Choose your answer
Was this exam question helpful?
Which row of the table correctly describes the link between outer shell electrons, Group number and Period number?
| outer electrons same as | group number same as | period number same as |
A | group number | period number | outer electrons |
B | period number | group number | outer electrons |
C | group number | outer electrons | number of electron shells |
D | outer electrons | group number | number of electron shells |
Choose your answer
Was this exam question helpful?
Which row correctly describes the number of outer shell electrons and properties of isotopes?
| Number of electrons | Chemical properties |
A | same | same |
B | same | different |
C | different | same |
D | different | different |
Choose your answer
Was this exam question helpful?
Iridium has two isotopes, 191Ir and 193Ir. Calculate the relative atomic mass of iridium using the data given:
Mass number | 191 | 193 |
Percentage abundance | 62.7% | 37.3% |
191.2
191.6
191.7
192.5
Choose your answer
Was this exam question helpful?
A selenide ion has the following notation.
Which row correctly describes the subatomic particles in a selenide ion?
| Protons | Neutrons | Electrons |
A | 79 | 34 | 34 |
B | 34 | 45 | 34 |
C | 34 | 45 | 36 |
D | 45 | 34 | 32 |
Choose your answer
Was this exam question helpful?
Copper has two naturally occurring isotopes, 63Cu and 65Cu. The relative abundance of 63Cu is 69%.
What is the relative atomic mass of copper?
63.6
64.0
64.4
65.0
Choose your answer
Was this exam question helpful?