Which of the following compounds are formed by covalent bonding?
1 | Potassium fluoride |
2 | Propane |
3 | Carbon dioxide |
4 | Lithium bromide |
2 and 3
2 and 4
1, 2 and 3
1 and 4
Was this exam question helpful?
Exam code: 0620 & 0971
Select a download format for Simple Molecules & Covalent Bonds
Select an answer set to view for
Simple Molecules & Covalent Bonds
Smart practice available on this topic
Answer questions with adaptive difficulty and personalised feedback
Which of the following compounds are formed by covalent bonding?
1 | Potassium fluoride |
2 | Propane |
3 | Carbon dioxide |
4 | Lithium bromide |
2 and 3
2 and 4
1, 2 and 3
1 and 4
Choose your answer
Was this exam question helpful?
Which row correctly describes a simple covalent compound?
| low melting point | high boiling point |
A | ||
B | ||
C | ||
D |
Choose your answer
Was this exam question helpful?
Which statement about bonding is not correct?
Covalent bonding involves non metal elements.
Covalent bonding involves electron sharing.
Covalent bonding involves electron transfer.
Covalent bonding does not involve metal elements.
Choose your answer
Was this exam question helpful?
Ammonia is a covalent compound consisting of one nitrogen atom bonded to three hydrogen atoms.
Which statement is correct for a molecule of ammonia?
The number of shared pairs of electrons is five
The number of shared pairs of electrons is three
There are single covalent bonds between the hydrogen atoms.
Each hydrogen atom donates one pair of electrons to the central nitrogen atom.
Choose your answer
Was this exam question helpful?
The structure of propane is shown below
Which correctly explains why propane can not conduct electricity?
There are weak intermolecular forces between molecules of propane
The covalent bonds in propane are strong
There are no free electrons able to move and carry a charge
Propane has a low boiling point
Choose your answer
Was this exam question helpful?
Lithium hydroxide reacts with hydrochloric acid to form lithium chloride and water.
Which row describes correctly the properties of the products and the final reaction mixture?
melting point of lithium chloride | melting point of water | electricity conduction of the reaction mixture | |
A | high | high | good |
B | high | low | good |
C | low | high | poor |
D | low | low | poor |
Choose your answer
Was this exam question helpful?
Atoms P and Q have electronic structures as shown in the diagram. They react together to form a covalent compound.

What is the formula of the compound?
P2Q3
P3Q
P3Q2
PQ4
Choose your answer
Was this exam question helpful?
The atom of element Z has 6 electrons in its outer shell. How would this element react?
It shares two electrons with four from another atom to form two covalent bonds
It shares four electrons with four from another atom to form four covalent bonds
It shares four electrons with two from another atom to form two covalent bonds
It shares two electrons with two from another atom to form two covalent bonds
Choose your answer
Was this exam question helpful?
CH4, H2O and HCl are covalent compounds.
Which atoms in these compounds do not use all of their outer shell electrons in bonding?
C and O
C and H
Cl and O
Cl and C
Choose your answer
Was this exam question helpful?
The electronic configurations of four different atoms are shown below.

Which atoms form covalent compounds?
W and X
W, X and Y
W and Z
X, Y and Z
Choose your answer
Was this exam question helpful?
Which of the following molecules contains only single covalent bonds?
N2
F2
O2
CO2
Choose your answer
Was this exam question helpful?
Diagrams 1 and 2 show the electron arrangements in molecules of water and methane.

Which row describes the diagrams?
water diagram | methane diagram | |
A | correct | incorrect |
B | correct | correct |
C | incorrect | incorrect |
D | incorrect | correct |
Choose your answer
Was this exam question helpful?
Carbon dioxide is a simple molecular substance in which the molecules are formed by covalent bonding between one carbon atom and two oxygen atoms.
Which dot and cross diagram below shows the correct outer shell arrangement of electrons in a molecule of CO2?

Choose your answer
Was this exam question helpful?
A student has drawn a dot and cross diagram to represent a molecule of ethene, C2H4, as shown below.

What is wrong with the students drawing?
The number of electrons shared between the carbon atoms is incorrect.
The number of electrons shared between each carbon and hydrogen is incorrect.
There is nothing wrong with the drawing.
The total number of electrons shared is incorrect.
Choose your answer
Was this exam question helpful?
Dot and cross diagrams for four covalent molecules are shown below.
Which diagram is incorrect?

Choose your answer
Was this exam question helpful?
Which statement about methane is correct?
It has a high melting point due to strong covalent bonds between atoms
It has a low melting point due to weak covalent bonds between the molecules
It has a low boiling point due to weak intermolecular forces between molecules
It has a high boiling point because of strong intermolecular forces between molecules
Choose your answer
Was this exam question helpful?
A covalent molecule M contains a total of four shared electrons.
What is M?
Carbon dioxide
Oxygen
Nitrogen
Methane
Choose your answer
Was this exam question helpful?
The displayed formula of ethanoic acid is shown.
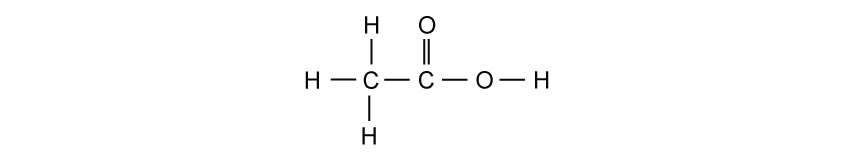
Which row shows the total number of shared electrons and the total number of non-bonding electrons in one molecule of ethanoic acid?
total number of shared electrons | total number of non-bonding electrons | |
A | 14 | 0 |
B | 14 | 8 |
C | 16 | 0 |
D | 16 | 8 |
Choose your answer
Was this exam question helpful?