Atomic Number & The Periodic Table (AQA GCSE Combined Science: Synergy: Physical Sciences): Revision Note
Exam code: 8465
Written by: Stewart Hird
Updated on
Arrangement by atomic number
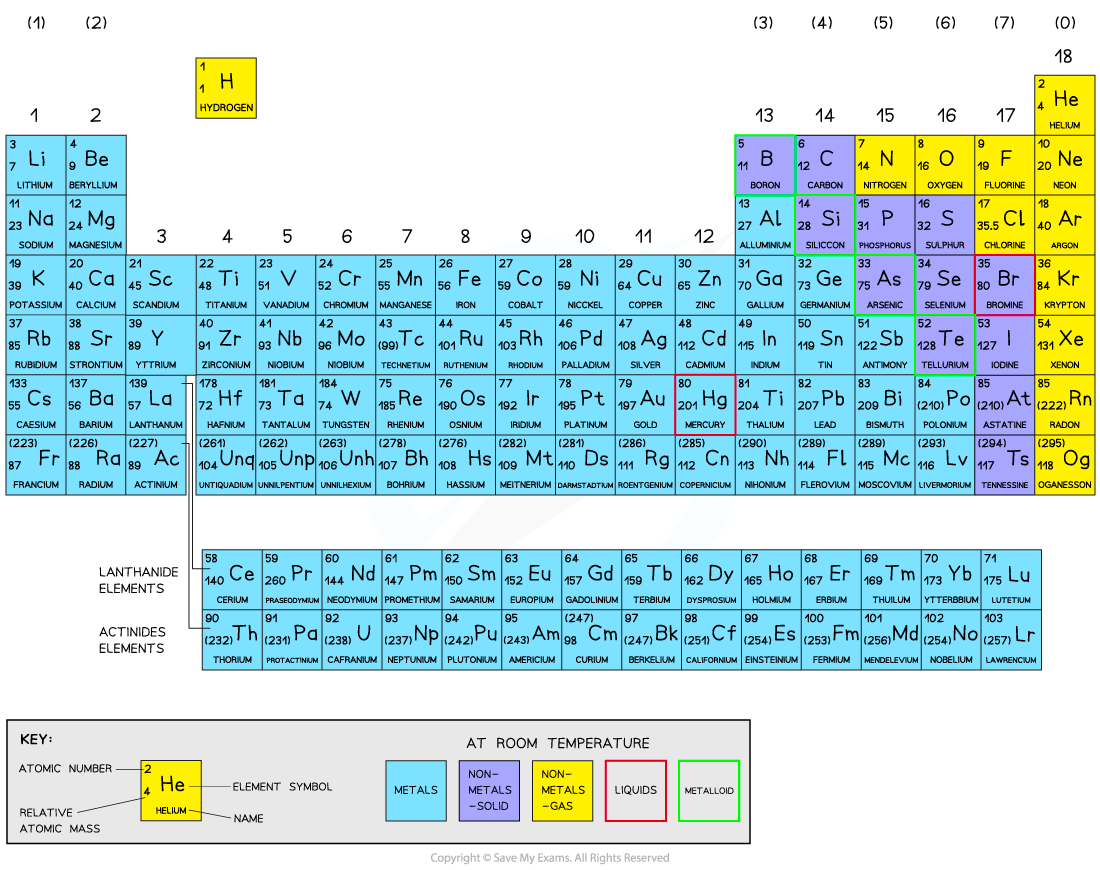
Over 100 chemical elements have been isolated and identified
Elements are arranged on the periodic table in order of increasing atomic number
Each element has one proton more than the element preceding it
This groups elements with similar chemical properties into the same vertical columns
Groups and periods
The table is arranged in:
Vertical columns called groups
Horizontal rows called periods
Groups
The groups are numbered from 1 – 7, with a final group called Group 0 (instead of Group 8)
The group number indicates how many electrons an atom has in its outer shell
For example, elements in Group 2 have 2 electrons in their outer shell
Periods
The periods are numbered from 1 - 7
The period number indicates the number of electron shells that an atom has
For example, elements in Period 3 have 3 electron shells
How does the electronic structure of an element relate to its location in the Periodic Table?
There is a clear relationship between the electronic configuration and how the Periodic Table is designed
The number of notations in the electronic configuration tells us the number of occupied shells
This tells us what period an element is in
The last notation shows the number of outer electrons the atom has
This tells us the group an element is in
Group number | Number of outer electrons | Example electronic configuration |
|---|---|---|
1 | 1 | 2,1 |
2 | 2 | 2,8,8,2 |
3 | 3 | 2,8,3 |
4 | 4 | 2,4 |
5 | 5 | 2,8,5 |
6 | 6 | 2,6 |
7 | 7 | 2,8,7 |
0 | 8 (or 2 for the first period) | 2 for helium 2,8 for neon |
Examiner Tips and Tricks
The atomic number is unique to each element and could be considered as an element's “fingerprint”.
The number of electrons changes during chemical reactions, but the atomic number does not change.
Electrons and shells
An element's electronic configuration determines its position in the periodic table
The number of occupied electron shells tells you which period the element is in
The number of electrons in the outer shell tells you which group the element is in
Elements in the same group have the same number of outer shell electrons
This is why they have similar chemical properties
We can represent the structure of the atom in two ways:
Using diagrams called electron shell diagrams
By writing out a notation called the electronic configuration
Electron shell diagrams
Electrons orbit the nucleus in shells (or energy levels) and each shell has a different amount of energy associated with it
The further away from the nucleus, the more energy a shell has
Electrons fill the shell closest to the nucleus
When a shell becomes full of electrons, additional electrons have to be added to the next shell
The first shell can hold 2 electrons
The second shell can hold 8 electrons
For this course, a simplified model is used that suggests that the third shell can hold 8 electrons
For the first 20 elements, once the third shell has 8 electrons, the fourth shell begins to fill
The outermost shell of an atom is called the valence shell and an atom is much more stable if it can manage to completely fill this shell with electrons

Electronic configuration
The arrangement of electrons in shells can also be explained using numbers
Instead of drawing electron shell diagrams, the number of electrons in each electron shell can be written down, separated by commas
This notation is called the electronic configuration (or electronic structure)
The electronic structure of carbon is 6 electrons, 2 in the 1st shell and 4 in the 2nd shell
So its electronic configuration is 2,4
Electronic configurations can also be written for ions
E.g. A sodium atom has 11 electrons, a sodium ion has lost one electron, therefore has 10 electrons; 2 in the first shell and 8 in the 2nd shell
Its electronic configuration is 2,8
Diagram showing the relationship between the electronic configurations

Period: The red numbers at the bottom show the number of notations
The number of notations is 3
Therefore chlorine has 3 occupied shells
Group: The last notation, in this case 7
This means that chlorine has 7 electrons in its outer shell
Chlorine is therefore in Group 7
The Periodic Table showing the location of chlorine

Examiner Tips and Tricks
All of the inner shells will be full. Electron transfer occurs with electrons from the outer shell only.
Mendeleev and the Periodic Table
Early Periodic Tables
Before subatomic particles were discovered, scientists arranged elements in order of atomic mass
Patterns appeared at regular intervals
This is why we use the term "periodic"
Some elements were placed in the wrong group because chemical properties were overridden by strict mass ordering
Tables were incomplete, with gaps that couldn't be filled by known elements
Mendeleev's arrangement
In 1869, Dmitri Mendeleev organised elements into columns based on their chemical properties
They were then further arranged horizontally by increasing atomic mass
Where elements didn't fit, he left gaps rather than forcing them into place
He predicted that undiscovered elements would eventually fill the gaps
Mendeleev's Periodic Table

When those elements were later discovered and matched his predictions, it confirmed his approach
Isotopes and atomic number
Isotopes were unknown in Mendeleev's time
Since the same element can have atoms with different masses, ordering by atomic mass alone created unavoidable anomalies
Once subatomic particles were discovered, atomic number was calculated for each element
Reordering by atomic number (rather than mass) resolved the anomalies and gave us the modern periodic table
Examiner Tips and Tricks
Mendeleev's key insight was leaving gaps rather than forcing elements into the wrong position. This allowed him to predict the existence and properties of then-unknown elements.

Unlock more, it's free!
Was this revision note helpful?
