The graph below shows a reaction profile for the reaction between methane and oxygen.

What does the label X represent?
Activation energy
Overall energy change
Reactants
Progress of the reaction
Was this exam question helpful?
Exam code: 5070
Select a download format for 5.1 Exothermic & Endothermic Reactions
Select an answer set to view for
5.1 Exothermic & Endothermic Reactions
The graph below shows a reaction profile for the reaction between methane and oxygen.

What does the label X represent?
Activation energy
Overall energy change
Reactants
Progress of the reaction
Choose your answer
Was this exam question helpful?
What is the reading on the following thermometer?

28.3 oC
28.6 oC
29.2 oC
28.5 oC
Choose your answer
Was this exam question helpful?
A student adds a small amount of ammonium chloride to a beaker of water. The temperature of the water decreases from 21 oC to 17 oC.
Which type of reaction has occurred and why?
| type of reaction | reason |
A | exothermic | heat is released |
B | exothermic | heat is absorbed |
C | endothermic | heat is released |
D | endothermic | heat is absorbed |
Choose your answer
Was this exam question helpful?
The energy level diagram below shows the relative energies of the reactants and products in a reaction.

Which row correctly describes the type of reaction and corresponding energy change?
| type of reaction | energy change |
A | endothermic | heat is released |
B | endothermic | heat is absorbed |
C | exothermic | heat is released |
D | exothermic | heat is absorbed |
Choose your answer
Was this exam question helpful?
Calcium and hydrochloric acid react to produce calcium chloride and hydrogen gas. The energy level diagram for the reaction is shown below.

Which statement about this reaction is not correct?
The temperature of the surroundings increases.
The reaction is endothermic.
The products have less energy than the reactants.
Energy is released during the reaction.
Choose your answer
Was this exam question helpful?
The reaction pathway diagram for a reaction, with and without a catalyst, is shown.
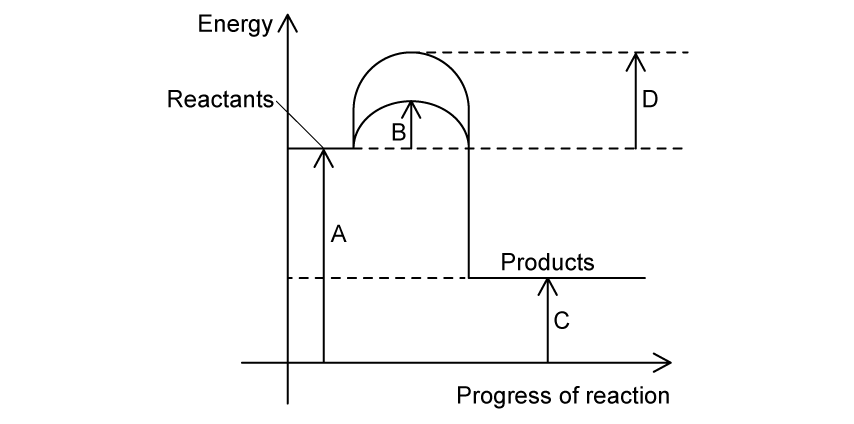
Which letter represents the activation energy of the catalysed reaction?
Choose your answer
Was this exam question helpful?
Which of the following processes is endothermic?
Reacting sodium with water.
The use of petrol in an engine.
Distilling crude oil.
Burning fossil fuels.
Choose your answer
Was this exam question helpful?
In all chemical reactions, bonds in the reactant species are broken and new bonds are formed in the products. Which statement is correct?
Bond breaking requires energy and is thus an exothermic process.
Bond breaking requires energy and is thus an endothermic process.
Bond breaking releases energy and is thus an exothermic process.
Bond breaking releases energy and is thus an endothermic process.
Choose your answer
Was this exam question helpful?
Which is true about chemical energy changes?
| Reaction type | Energy flows | Temperature of surroundings |
A | exothermic | to the surroundings | increases |
B | exothermic | from the surroundings | decreases |
C | endothermic | to the surroundings | decreases |
D | endothermic | from the surroundings | increases |
Choose your answer
Was this exam question helpful?
The reaction profile for making nitrogen monoxide, NO, is shown below:

What is the activation energy for this reaction?
+140 kJ
+180 kJ
+320 kJ
-330 kJ
Choose your answer
Was this exam question helpful?
Endothermic reactions can be used in emergency sports injury treatment packs.
What is true about these packs?
Absorb energy from the surroundings and transfer it to the injury
Can be re-used
Usually contain ammonium nitrate and water in separate compartments
Cause the temperature of the surroundings to increase
Choose your answer
Was this exam question helpful?
An endothermic reaction has an activation energy of x.
Which reaction pathway diagram is correct for this reaction?

Choose your answer
Was this exam question helpful?
The formation of liquid water from hydrogen and oxygen may occur in three stages.
1 | 2H2(g) + O2(g) → 4H(g) + 2O(g) |
2 | 4H(g) + 2O(g) → 2H2O(g) |
3 | 2H2O(g) → 2H2O(l) |
Which stages are endothermic?
1, 2 and 3
1 only
1 and 3
2 only
Choose your answer
Was this exam question helpful?
Chlorine and ethane react together in an exothermic reaction to produce chloroethane and hydrogen chloride. The equation for the reaction is shown below.
C2H6 + Cl2 → C2H5Cl + HCl
The table below shows the relevant bond energies.
bond | bond energy / (kJ / mol) |
CーCl | + 340 |
CーC | + 350 |
CーH | + 410 |
ClーCl | + 240 |
HーCl | + 430 |
What is the energy change for the reaction between chlorine and ethane?
-120 kJ / mol
-1020 kJ / mol
+120 kJ / mol
+1020 kJ / mol
Choose your answer
Was this exam question helpful?
The production of ammonia involves the exothermic reaction between nitrogen and hydrogen. The equation for the reaction is shown.
N2 + 3 H2 → 2 NH3
The table shows the bond energies.
bond | bond energy / (kJ / mol) |
NーH | +390 |
HーH | +436 |
N☰N | +945 |
What is the energy released during the reaction?
-230 kJ / mol
-1025 kJ / mol
-87 kJ / mol
+87 kJ / mol
Choose your answer
Was this exam question helpful?
Chlorine and hydrogen react in an exothermic reaction producing hydrogen chloride. The equation for the reaction is shown.
Cl2 + H2 → 2 HCl
The table shows some of the bond energies involved. The total energy change for this reaction is -184 kJ / mol.
bond | bond energy / (kJ / mol) |
HーCl | +430 |
HーH | +436 |
ClーCl | - |
What is the energy of the Cl─Cl bond?
-170 kJ / mol
+240 kJ / mol
+170 kJ / mol
-240 kJ / mol
Choose your answer
Was this exam question helpful?
Methane combusts in oxygen as shown producing an energy change of -890 kJ / mol.
CH4 + 2O2 → CO2 + 2H2O
The table shows some of the bond energies involved.
bond | bond energy / (kJ / mol) |
HーO | +460 |
O=O | +496 |
CーH | +410 |
C=O | - |
What is the energy of the C=O bond?
-155 kJ / mol
+333 kJ / mol
-840 kJ / mol
+841 kJ / mol
Choose your answer
Was this exam question helpful?
An energy level diagram is shown below.

Which row is correct for the points labelled 1, 2 and 3?
| 1 | 2 | 3 |
A | activation energy | energy change | reactants |
B | energy change | activation energy | products |
C | energy change | activation energy | reactants |
D | activation energy | energy change | products |
Choose your answer
Was this exam question helpful?