Isotopes (AQA GCSE Combined Science: Synergy: Life & Environmental Sciences): Revision Note
Exam code: 8465
Written by: Stewart Hird
Updated on
Mass Number
What is the mass number of an atom?
The mass number (or nucleon number) is the total number of protons and neutrons in the nucleus of an atom
The symbol for this number is A
The nucleon number minus the proton number gives you the number of neutrons of an atom
Note that protons and neutrons can collectively be called nucleons
The atomic number and mass number for every element is on the periodic table

Diagram showing the notation used on the periodic table
Isotopes
What is an isotope?
Isotopes are atoms of the same element that have the same number of protons but a different number of neutrons
This means isotopes have the same atomic number but different mass numbers
The symbol for an isotope is the chemical symbol (or word) followed by a dash and then the mass number
So, C-14 is the isotope of carbon which contains 6 protons and 6 electrons
But, the 14 signifies that it has 8 neutrons (14 - 6 = 8)
It can also be written as 14C
Isotopes display the same chemical characteristics
This is because they have the same number of electrons in their outer shells, and this is what determines their chemistry
The difference between isotopes is their neutrons, which are neutral particles that add mass only
Table to show the structure of isotopes of hydrogen

The atomic structure and symbols of the three isotopes of hydrogen
Examiner Tips and Tricks
Isotopes of the same element always have the same atomic number.
For atoms to be isotopes of each other, they must both be from the same element. For example:
C-13 and C-14 are isotopes
C-13 and H-2 are not isotopes
Representation
Atoms are represented using the following notation, showing the mass number and atomic number alongside the chemical symbol:
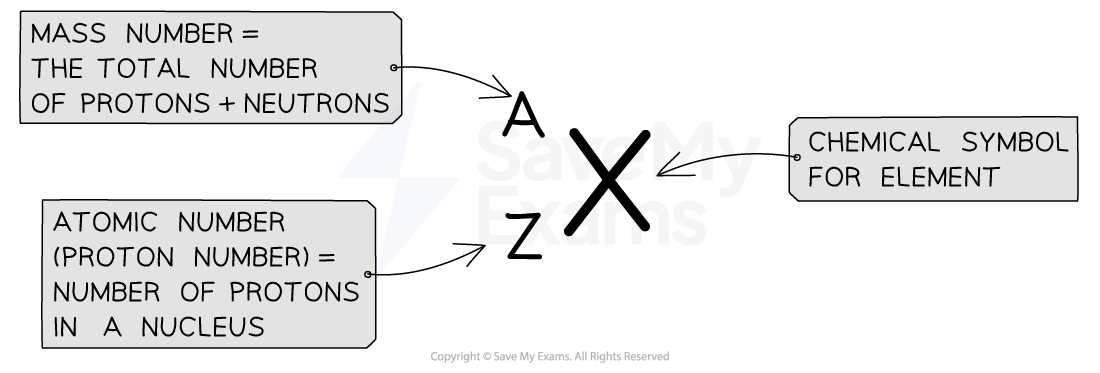
For example:
Element | Chemical symbol |
|---|---|
Carbon | |
Oxygen | |
Sodium |
From this notation, the number of protons, electrons and neutrons can be worked out:
Number of protons = atomic number
Number of electrons = atomic number (for a neutral atom)
Number of neutrons = mass number − atomic number
Worked Example
The chemical symbol for an atom of beryllium is:

Calculate the number of protons, electrons and neutrons.
Answer:
Protons = 4
Electrons = 4
Neutrons = 9 − 4 = 5
For ions, the electron count must be adjusted for the charge:
Negative ions
Have gained electrons
Add the charge value to the electron count
Positive ions
Have lost electrons
Subtract the charge value from the electron count
Worked Example
The chemical symbol for a fluoride ion is:
Calculate the number of protons, electrons and neutrons.
Answer:
Protons = 9
Electrons = 9 + 1 = 10 (gained one electron due to −1 charge)
Neutrons = 19 − 9 = 10
Worked Example
The chemical symbol for a magnesium ion is:
Calculate the number of protons, electrons and neutrons.
Answer:
Protons = 12
Electrons = 12 -2 = 10 (lost two electrons due to +2 charge)
Neutrons = 24 - 12 = 12
Examiner Tips and Tricks
The mass number is always the larger of the two numbers in the notation. Use mass number − atomic number to find neutrons. This is a very common exam calculation.

Unlock more, it's free!
Was this revision note helpful?
