Half-Life (AQA GCSE Combined Science: Synergy: Life & Environmental Sciences): Revision Note
Exam code: 8465
Written by: Ashika
Updated on
Random Nature of Decay
It cannot be predicted when a particular unstable nucleus will decay
This is because radioactive decay is a random process, this means that:
There is an equal probability of any nucleus decaying
It cannot be known which particular nucleus will decay next
It cannot be known at what time a particular nucleus will decay
The rate of decay is unaffected by the surrounding conditions
It is only possible to estimate the probability of a nuclei decaying in a given time period
For example, a researcher might take some readings of background radiation
If the researcher reset the counter to zero, waited one minute and then took the count
reading and repeated the procedure, they might obtain results such as:
32 11 25 16 28
The readings don't appear to follow a particular trend
This happens because of the randomness of radioactive decay
Dice Analogy
An analogy is a way of understanding an idea by using a different but similar situation
Rolling dice is a good analogy of radioactive decay because it is similar to the random nature of radioactive decay

A dice roll is a random process because you don't know when you will roll a particular value. However, you can determine the probability of a particular result
Imagine someone rolling a dice and trying to get a ‘6’
Each time they roll, they do not know what the result will be
But they know there is a 1/6 probability that it will be a 6
If they were to roll the dice 1000 times, it would be very likely that they would roll a 6 at least once
The random nature of radioactive decay can be demonstrated by observing the count rate of a Geiger-Muller (GM) tube
When a GM tube is placed near a radioactive source, the counts are found to be irregular and cannot be predicted
Each count represents a decay of an unstable nucleus
These fluctuations in count rate on the GM tube provide evidence for the randomness of radioactive decay

The variation of count rate over time of a sample radioactive gas. The fluctuations show the randomness of radioactive decay
Half-Life
It is impossible to know when a particular unstable nucleus will decay
But the rate at which the activity of a sample decreases can be known
This is known as the half-life
Half-life is defined as:
The time it takes for the number of nuclei of a sample of radioactive isotopes to decrease by half
OR
The time it takes for the count rate of a sample to fall to half its initial level
In other words, the time it takes for the activity of a sample to fall to half its original level
Count rate is the number of decays recorded each second by a detector, such as a Geiger–Müller (GM) tube
Different isotopes have different half-lives and half-lives can vary from a fraction of a second to billions of years in length
Using Half-life
Scientists can measure the half-lives of different isotopes accurately:
Uranium-235 has a half-life of 704 million years
This means it would take 704 million years for the activity of a uranium-235 sample to decrease to half its original amount
Carbon-14 has a half-life of 5700 years
So after 5700 years, there would be 50% of the original amount of carbon-14 remaining
After two half-lives, or 11 400 years, there would be just 25% of the carbon-14 remaining
With each half-life, the amount remaining decreases by half
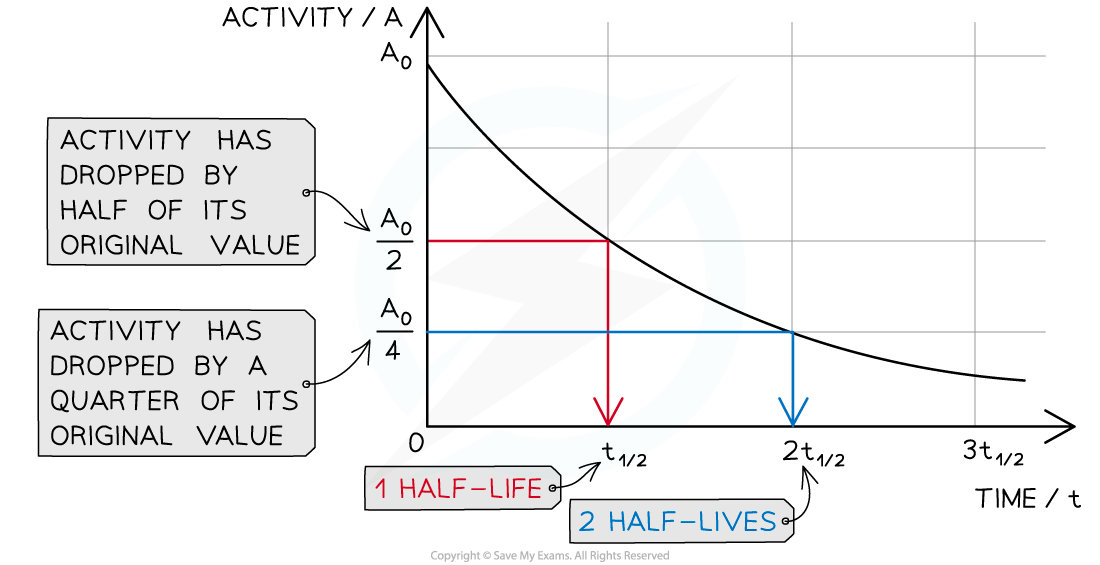
The diagram shows how the activity of a radioactive sample changes over time. Each time the original activity halves, another half-life has passed
The time it takes for the activity of the sample to decrease from 100 % to 50 % is the half-life
It is the same length of time as it would take to decrease from 50 % activity to 25 % activity
The half-life is constant for a particular isotope
Calculating Half-Life
To calculate the half-life of a sample, the procedure is:
Measure the initial activity, A0, of the sample
Determine the half-life of this original activity
Measure how the activity changes with time
The time taken for the activity to decrease to half its original value is the half-life
Worked Example
The radioisotope technetium is used extensively in medicine. The graph below shows how the activity of a sample varies with time.

Determine the half-life of this material.
Answer:
Step 1: Draw lines on the graph to determine the time it takes for technetium to drop to half of its original activity

Step 2: Read the half-life from the graph
In the diagram above the initial activity, A0, is 8 × 107 Bq
The time taken to decrease to 4 × 107 Bq, or ½ A0, is 6 hours
The time taken to decrease to 2 × 107 Bq is 6 more hours
The time taken to decrease to 1 × 107 Bq is 6 more hours
Therefore, the half-life of this isotope is 6 hours
Worked Example
A particular radioactive sample contains 2 million un-decayed atoms. After a year, there are only 500 000 atoms left un-decayed. What is the half-life of this material?
Answer:
Step 1: Calculate how many times the number of un-decayed atoms has halved
There were 2 000 000 atoms to start with
1 000 000 atoms would remain after 1 half-life
500 000 atoms would remain after 2 half-lives
Therefore, the sample has undergone 2 half-lives
Step 2: Divide the time period by the number of half-lives
The time period is a year
The number of half-lives is 2
So two half-lives is 1 year, and one half-life is 6 months
Therefore, the half-life of the sample is 6 months
Calculating Radioactive Decay
Higher Tier Only
Half-life is the time it takes for the number of nuclei of a sample of radioactive isotopes to decrease by half
With each half-life, the activity of a sample decreases by half
The ratio of remaining radioactive nuclei to the decayed nuclei after a period of time can be calculated in different ways
Method 1: Halving Method
Determine the number of half-lives elapsed
Multiply the number 1 by half for each half-life elapsed
For example, if 4 half-lives have elapsed:
1 × ½ × ½ × ½ × ½ = 1 / 16
This is the same as a ratio of 1 remaining : 16 original nuclei, or 1:16
Method 2: Raising to a Power
Determine the number of half-lives elapsed
Use your calculator to raise ½ to the number of half-lives
For example, if 4 half-lives have elapsed:
(1/2)4 = 1/16
This is the same as a ratio of 1 remaining : 16 original nuclei, or 1:16
Worked Example
A radioactive sample has a half-life of 3 years. What is the ratio of decayed : remaining nuclei, after 15 years?
Answer:
Step 1: Calculate the number of half-lives
The time period is 15 years
The half-life is 3 years
15 ÷ 3 = 5
There have been 5 half-lives
Step 2: Raise 1/2 to the number of half-lives
(1/2)5 = 1/32
So 1/32 of the original nuclei are remaining
Step 3: Write the ratio correctly
If 1/32 of the original nuclei are remaining, then 31/32 must have decayed
Therefore, the ratio is 31 decayed : 1 remaining, or 31:1

Unlock more, it's free!
Was this revision note helpful?
