The Water Cycle (AQA GCSE Combined Science: Synergy: Life & Environmental Sciences): Revision Note
Exam code: 8465
Written by: Lára Marie McIvor
Updated on
The Water Cycle
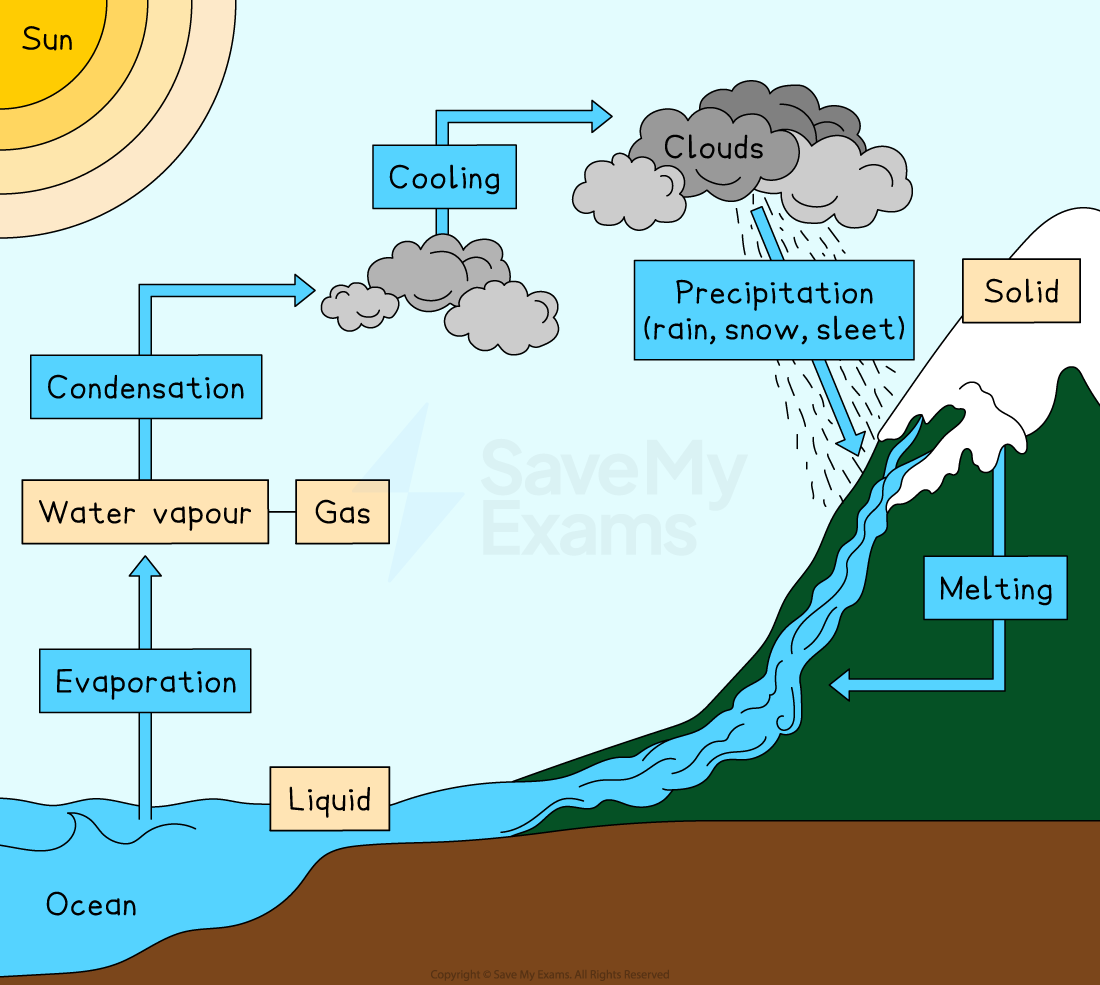
Water is found in three states across the Earth:
Solid: in glaciers and ice sheets
Liquid: in oceans, rivers, lakes and underground aquifers
Gas: as water vapour in the atmosphere
Water continuously cycles between these states through several processes
Evaporation
Energy from the Sun causes liquid water to evaporate from oceans, rivers and lakes, turning into water vapour in the atmosphere
As water vapour rises, the air cools
Condensation
Cooling water vapour condenses back into tiny liquid droplets, forming clouds
Precipitation
As water droplets in clouds get bigger and heavier, they fall back to Earth as precipitation
Precipitation can take the form of rain, sleet, snow or hail
Melting and freezing
Solid water in glaciers and ice sheets can melt, flowing as liquid into rivers and oceans
Under cold conditions, liquid water freezes back into a solid
Examiner Tips and Tricks
Make sure you can name all the key processes shown in water cycle diagrams:
Evaporation
Condensation
Precipitation
Melting
Freezing
Extended questions may ask you to trace the journey of water molecules between states. For example, from an iceberg to a lake.
Structure your answer as a sequence:
Solid iceberg
→ melts to liquid
→ evaporates to gas
→ rises and cools
→ condenses into cloud droplets
→ falls as precipitation into the lake.
Each state change and process should be named explicitly.
Why is water important?
Life on Earth depends on water, both on land and in the seas
Water as a solvent
Water acts as the solvent for chemical reactions in cells
Many substances dissolve in water, allowing biochemical reactions to take place
Water as a transport medium
Water transports dissolved compounds into and out of cells
It carries nutrients, waste products and other substances around organisms
Water as a reactant and product
Water is either a reactant or a product in many biochemical reactions, including:
Photosynthesis:
carbon dioxide + water → glucose + oxygen
Respiration:
glucose + oxygen → carbon dioxide + water
Digestion: large food molecules are broken down with water
Water as a habitat
Rivers, lakes and seas provide habitats for a huge variety of living organisms
Examiner Tips and Tricks
The importance of water has not appeared as a standalone explanation question in recent Synergy past papers.
However, water's role as a reactant is regularly tested through photosynthesis equation completion: carbon dioxide + water → glucose + oxygen.
If asked about water's importance more broadly, give specific examples and name the biochemical reactions rather than just saying "water is important for life."

Unlock more, it's free!
Was this revision note helpful?
