Pollutants That Affect Air Quality (AQA GCSE Combined Science: Synergy: Life & Environmental Sciences): Revision Note
Exam code: 8465
Written by: Stewart Hird
Updated on
Combustion of fossil fuels
The combustion of fossil fuels is a major source of atmospheric pollution
Fossil fuels include coal, oil, natural gas, oil shales, and tar sands
These fuels are mainly hydrocarbons, containing only carbon and hydrogen
Fuels may also contain small amounts of sulfur as impurities
When burned, the fuels release energy but also a variety of pollutants that can harm the environment and human health
Combustion products
Burning fossil fuels releases several gases into the atmosphere:
Carbon dioxide (CO2) – produced during complete combustion
Water vapour (H2O) – also from complete combustion
Carbon monoxide (CO) – produced during incomplete combustion when oxygen is limited
Oxides of nitrogen (NO and NO2) – formed from nitrogen and oxygen in the air at high temperatures in engines
Sulfur dioxide (SO2) – produced from sulfur impurities in the fuel
Solid particles and unburned hydrocarbons can also be released, forming soot (carbon particulates)
Complete vs incomplete combustion
Complete combustion happens when there is plenty of oxygen:
hydrocarbon + oxygen → carbon dioxide + water
example: C3H8 + 5O2 → 3CO2 + 4H2O
Incomplete combustion happens when there is not enough oxygen
hydrocarbon + oxygen → carbon monoxide + water
example: 2CH4 + 3O2 → 2CO + 4H2O
Incomplete combustion is common in engines and poorly ventilated stoves
Formation of key pollutants
Carbon monoxide (CO)
Carbon monoxide is produced when fuels burn with limited oxygen
The carbon in the fuel is only partially oxidised
For example:
2CH4 + 3O2 → 2CO + 4H2O
Soot (carbon particles)
Soot forms when there is very little oxygen, causing hydrocarbons to break down without fully combusting
For example:
C8H18 + 8½O2 → 8C (soot) + 9H2O
Sulfur dioxide (SO2)
Some fossil fuels contain sulfur as an impurity
During combustion, this sulfur reacts with oxygen to form sulfur dioxide:
S + O2 → SO2
Oxides of nitrogen (NO and NO2)
Oxides of nitrogen form when nitrogen and oxygen from the air react at the extreme temperatures achieved in car engines and power stations.
For example:
N2 + O2 → 2NO
2NO + O2 → 2NO2
Examiner Tips and Tricks
The equation for formation of nitrogen dioxide has also appeared in past papers, so it is worth being able to write and balance it:
N2 + 2O2 → 2NO2
Carbon monoxide and health
Carbon monoxide is an extremely poisonous gas as it combines with haemoglobin in blood and prevents it from carrying oxygen
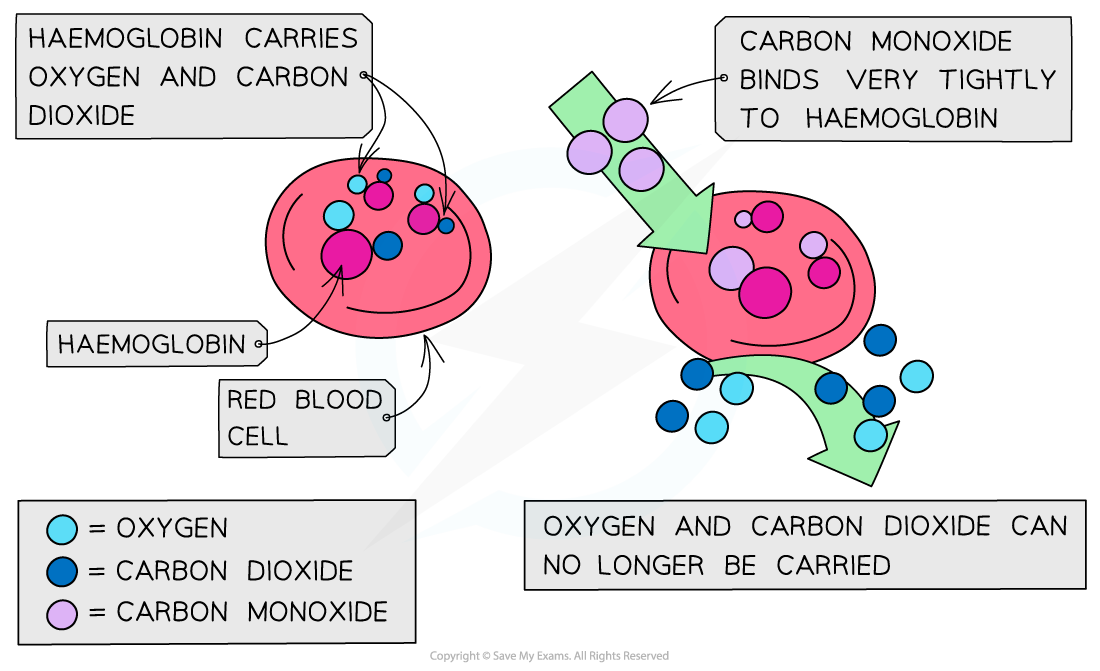
It is particularly dangerous as it is colourless and odourless
This makes it difficult to detect carbon monoxide
At low concentrations, symptoms include:
Faster heart rate (to compensate for reduced oxygen delivery to cells)
Dizziness
Tiredness
At high concentrations, cells receive insufficient oxygen for respiration and release less energy, causing loss of consciousness, coma or death
Examiner Tips and Tricks
Carbon monoxide questions frequently ask you to explain why CO increases heart rate:
CO binds to haemoglobin
→ red blood cells carry less oxygen
→ the heart beats faster to compensate.
Distinguish clearly between low-dose effects (faster heart rate, dizziness, tiredness) and high-dose effects (less energy released by cells → loss of consciousness).
Acid rain
Acid rain is commonly caused by:
Sulfur dioxide, SO2
Oxides of nitrogen, NOx
Sulfur dioxide
Sulfur dioxide is a colourless, pungent-smelling gas produced when fuels containing sulfur impurities are burned
The main sources are coal burned in power stations and diesel fuels used in ships and heavy vehicles
Sulfur dioxide dissolves in water in clouds to form sulfuric acid, which falls as acid rain
Oxides of nitrogen
Oxides of nitrogen form when nitrogen and oxygen react at the high temperatures reached in vehicle engines and power stations
They contribute to acid rain and photochemical smog, and cause respiratory problems, particularly for people with asthma
Effects of acid rain
Acid rain corrodes metal structures and buildings and statues made of carbonate rocks
It damages aquatic organisms in rivers, ponds and lakes
It pollutes crops and water supplies, and irritates the lungs, throat and eyes

Examiner Tips and Tricks
Greenhouse gases and atmospheric pollutants are not the same thing. A greenhouse gas causes global warming; a pollutant causes problems close to the ground such as smog, acid rain and respiratory damage.
Particulates
Particulates are tiny solid particles released by human activity
Sources include soot from diesel engines and dust from roads and industry
Soot forms during incomplete combustion when very little oxygen is available
Health and environmental effects
Larger particulates clump together to form soot
The soot then falls back to the ground and blackens buildings and statues, accelerating corrosion
Smaller particulates are inhaled and penetrate deep into the lungs
These smaller particulates cause damage that can lead to heart disease and lung cancer

Examiner Tips and Tricks
The body has defences against particulates: mucus secreted by the trachea and bronchi traps particles, and cilia sweep the mucus away from the lungs. This mechanism has been tested for up to 3 marks.
Particulates are sometimes classified by size in data questions. For example, PM2.5 refers to particles smaller than 2.5 micrometres, which are the most dangerous as they penetrate deepest into the lungs.

Unlock more, it's free!
Was this revision note helpful?
